Z. Kristallogr. NCS 219 (2004) 289-290
© by Oldenbourg Wissenschaftsverlag, M ü n c h e n
289
Crystal structure of diethyl re/-(3a/?,3b/?,6aS,7al?)-3b,6a,7,7a-tetrahydro- 3aH-cyclopenta-[l,2-J:3,4-rf']diisoxazole-3,6-dicarboxylate, C13H16N2O6
W. Frey, J . Y . Lee and V. Jäger*
Universität Stuttgart. Institut für Organische Chemie, Pfaffenwaldring 55, 70569 Stuttgart, Germany
Received March 30, 2004, accepted and available on-line July 28, 2004; CCDC no. 1267/1294
C13H16N2O6, orthorhombic,
P b c a(no. 61),
a= 9.8365(6) A, b = 12.0017(5) A, c = 23.920(1) A, V= 2823.8 A
3, Z = 8,
R g i( F )
= 0.076,
w R r e f f F2)= 0.238,
T =293 K.
Source of material
The title compound was obtained by heating ethyl re/-(3aS,6a5)- 4,6a-dihydro-3a//-cyclopenta[i/]isoxazole-3-carboxylate [1] with ethyl chlorooximidoacetate [2] in toluene under reflux [1,3]. The two diastereoisomers (ratio 52:48) were separated by column chromatography [4], whereby crystallization of the major isomer from petroleum ether furnished the title compound in the form of colorless crystals (m.p. 374—375 K, dec.).
Discussion
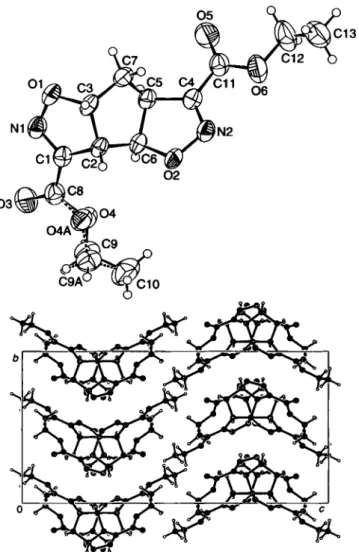
The crystal structure shows the
a n t iorientation of the tricyclic core. One of the ethyl ester side-chains is disordered with a popu- lation ratio of approx. 5:1. The isoxazoline moiety to which this ester function binds is nearly planar. The other isoxazoline moi- ety and the central cyclopentane show envelope conformations.
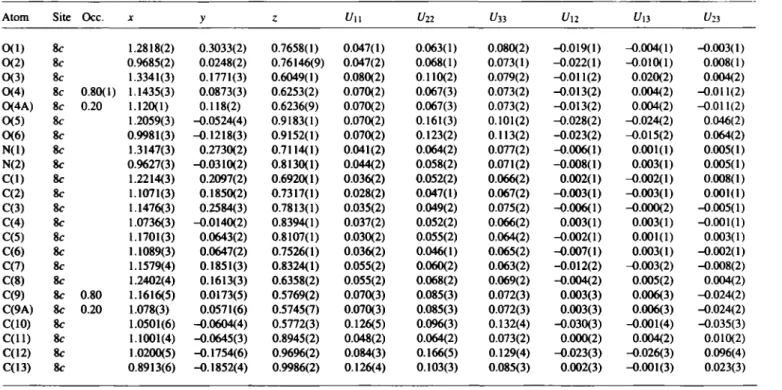
The second ethyl ester side-chain shows a strong librational be- haviour indicated by large displacement parameters which can- not be resolved into preferred positions (figure, top). In the cell plot (figure, bottom) we observe layers along the c-axis built up by the ester functions and the tricyclic ring systems.
Table 1. Data collection and handling.
colorless needle,
size 0.08 x 0.10 x 0.85 mm Cu Ka radiation (1.54178 A) 9.49 cm- 1
Siemens P4, <0 136°
3101,2393 A>bs > 2 of/obs), 1577 200
SHELXS-97 [5], SHELXL-97 [6]
Atom Site Occ. X y z Uis0
H(2) 8c 1.0185 0.2060 0.7162 0.057
H(3) 8c 1.0815 0.3185 0.7870 0.063
H(5) 8c 1.2641 0.0371 0.8110 0.059
H(6) 8c 1.1592 0.0159 0.7271 0.059
H(7A) 8c 1.0775 0.1932 0.8555 0.071
H(7B) 8c 1.2371 0.2049 0.8544 0.071
H(9A) 8c 0.80 1.1603 0.0616 0.5430 0.091 H(9B) 8c 0.80 1.2477 -0.0220 0.5789 0.091 H(9C) 8c 0.20 0.9973 0.0932 0.5601 0.091 H(9D) 8c 0.20 1.1489 0.0673 0.5466 0.091
H(10A) 8c 1.0575 -0.1093 0.5456 0.177
H(10B) 8c 0.9658 -0.0203 0.5753 0.177
H(10C) 8c 1.0526 -0.1034 0.6111 0.177
H(12A) 8c 1.0593 -0.2487 0.9642 0.152
H(12B) 8c 1.0827 -0.1314 0.9917 0.152
H(13A) 8c 0.9056 -0.2201 1.0342 0.156
H(13B) 8c 0.8300 -0.2296 0.9767 0.156
H(13C) 8c 0.8532 -0.1124 1.0041 0.156
Table 2. Atomic coordinates and displacement parameters (in A ).
Crystal:
Wavelength:
f t -
Diffractometer, scan mode:
2&nux~
N(/iWJmeasured, N(hkl)unique:
Criterion for /obs, N ( h k l )
N(param)tt fined:
Programs:
* Correspondence author (e-mail: jager.ioc@po.uni-stuttgart.de)
290
C l 3 H l 6 N206Table 3. Atomic coordinates and displacement parameters (in Â2).
Atom Site Occ. X y z Un U22 i/33 Un Un Uiì
CKD 8c 1.2818(2) 0.3033(2) 0.7658(1) 0.047(1) 0.063(1) 0.080(2) -0.019(1) -0.004(1) -0.003(1) CX2) 8c 0.9685(2) 0.0248(2) 0.76146(9) 0.047(2) 0.068(1) 0.073(1) -0.022(1) -0.010(1) 0.008(1) CK3) 8c 1.3341(3) 0.1771(3) 0.6049(1) 0.080(2) 0.110(2) 0.079(2) -0.011(2) 0.020(2) 0.004(2) CK4) 8c 0.80(1) 1.1435(3) 0.0873(3) 0.6253(2) 0.070(2) 0.067(3) 0.073(2) -0.013(2) 0.004(2) -0.011(2) CK4A) 8c 0.20 1.120(1) 0.118(2) 0.6236(9) 0.070(2) 0.067(3) 0.073(2) -0.013(2) 0.004(2) -0.011(2) 0(5) 8c 1.2059(3) -0.0524(4) 0.9183(1) 0.070(2) 0.161(3) 0.101(2) -0.028(2) -0.024(2) 0.046(2) 0(6) 8c 0.9981(3) -0.1218(3) 0.9152(1) 0.070(2) 0.123(2) 0.113(2) -0.023(2) -0.015(2) 0.064(2) N(l) 8c 1.3147(3) 0.2730(2) 0.7114(1) 0.041(2) 0.064(2) 0.077(2) -0.006(1) 0.001(1) 0.005(1) N(2) 8c 0.9627(3) -0.0310(2) 0.8130(1) 0.044(2) 0.058(2) 0.071(2) -0.008(1) 0.003(1) 0.005(1) C(l) 8c 1.2214(3) 0.2097(2) 0.6920(1) 0.036(2) 0.052(2) 0.066(2) 0.002(1) -0.002(1) 0.008(1) C(2) 8c 1.1071(3) 0.1850(2) 0.7317(1) 0.028(2) 0.047(1) 0.067(2) -0.003(1) -0.003(1) 0.001(1) C(3) 8c 1.1476(3) 0.2584(3) 0.7813(1) 0.035(2) 0.049(2) 0.075(2) -0.006(1) -0.000(2) -0.005(1) C(4) 8c 1.0736(3) -0.0140(2) 0.8394(1) 0.037(2) 0.052(2) 0.066(2) 0.003(1) 0.003(1) -0.001(1) C(5) 8c 1.1701(3) 0.0643(2) 0.8107(1) 0.030(2) 0.055(2) 0.064(2) -0.002(1) 0.001(1) 0.003(1) C(6) 8c 1.1089(3) 0.0647(2) 0.7526(1) 0.036(2) 0.046(1) 0.065(2) -0.007(1) 0.003(1) -0.002(1) C(7) 8c 1.1579(4) 0.1851(3) 0.8324(1) 0.055(2) 0.060(2) 0.063(2) -0.012(2) -0.003(2) -0.008(2) C(8) 8c 1.2402(4) 0.1613(3) 0.6358(2) 0.055(2) 0.068(2) 0.069(2) -0.004(2) 0.005(2) 0.004(2) C(9) 8c 0.80 1.1616(5) 0.0173(5) 0.5769(2) 0.070(3) 0.085(3) 0.072(3) 0.003(3) 0.006(3) -0.024(2) C(9A) 8c 0.20 1.078(3) 0.0571(6) 0.5745(7) 0.070(3) 0.085(3) 0.072(3) 0.003(3) 0.006(3) -0.024(2) C(10) 8c 1.0501(6) -0.0604(4) 0.5772(3) 0.126(5) 0.096(3) 0.132(4) -0.030(3) -0.001(4) -0.035(3) C ( l l ) 8c 1.1001(4) -0.0645(3) 0.8945(2) 0.048(2) 0.064(2) 0.073(2) 0.000(2) 0.004(2) 0.010(2) C(12) 8c 1.0200(5) -0.1754(6) 0.9696(2) 0.084(3) 0.166(5) 0.129(4) -0.023(3) -0.026(3) 0.096(4) C(13) 8c 0.8913(6) -0.1852(4) 0.9986(2) 0.126(4) 0.103(3) 0.085(3) 0.002(3) -0.001(3) 0.023(3)
Acknowledgments. We are grateful to Fonds der Chemischen Industrie and Bayer AG for financial support.
References
1. Lee, J.-Y.: Synthese neuartiger Furanomycin-Analoga und Versuche zur Synthese von Diaminodicaibonsäuren über Bis(isoxazoline). Disserta- tion. Universität Stuttgart, Germany, in preparation.
2. Skinner, G. S.: Deaminization. m . Evidence of the Existence of Aliphatic Diazonium Salts From the Formation of Chlorooximino Compounds.
J. Am. Chem. Soc. 46 (1924) 731-741.
3. de Micheli, C.; Caldirola, P.; de Amici, M.: An Efficient Synthesis of Isoxazoles and Isoxazolines. Heterocycles 23 (1985) 2479-2482.
4. Frey, W.; Lee, J.-Y.; Jäger, V.: Crystal Structure of diethyl re/-(3a5, 4aS,7a/?,7b/?)-4,4a,7a,7b-tetrahydro-3atf-cyclopenta[l,2-ii:4,3-<i']-di- isoxazole-3,S-dicarboxylate, C13H16N2O6. Z. Kristallogr. NCS 213 (2004) 287-288.
5. Sheldrick, G. M.: SHELXS-97. Program for the Solution of Crystal Structures. University of Göttingen, Germany 1997.
6. Sheldrick, G. M.: SHELXL-97. Program for the Refinement of Crystal Structures. University of Göttingen, Germany 1997.