226 Zeitschrift für Kristallographie - New Crystal Structures 212
© by R. Oldenbourg Verlag, München 1997
Crystal structure of (35,4/?,l ^)-3-hydroxy-4-(l / ,2 / -dihydroxyethyl)-aze- tidin-2-one, C5H9NO4
S. Henkel, Β. Krämer and V. Jäger
Universität Stuttgart, Institut für Organische Chemie, Pfaffenwaldring 55, D-70569 Stuttgart, Germany
Received August 16, 19%, CSD-No. 402601
Table 1. Parameters used for the X-ray data collection
Crystal: colorless block, size 0.2 χ 0.4 χ 0.7 mm Wavelength: Mo Ka radiation (0.71073 Â)
μ: 1.26 cm"1
Diffractometer. Nicolet P3
Scan mode: Wyckoff
Tmeasuremeru'·
293 Κ20max: 60°
NihkOuniqur-
1077Criterion for /0:
¡o >2
σ(/ο) Ν (param)refintd'· 120Programs: SHELXS-86, SHELXL-93
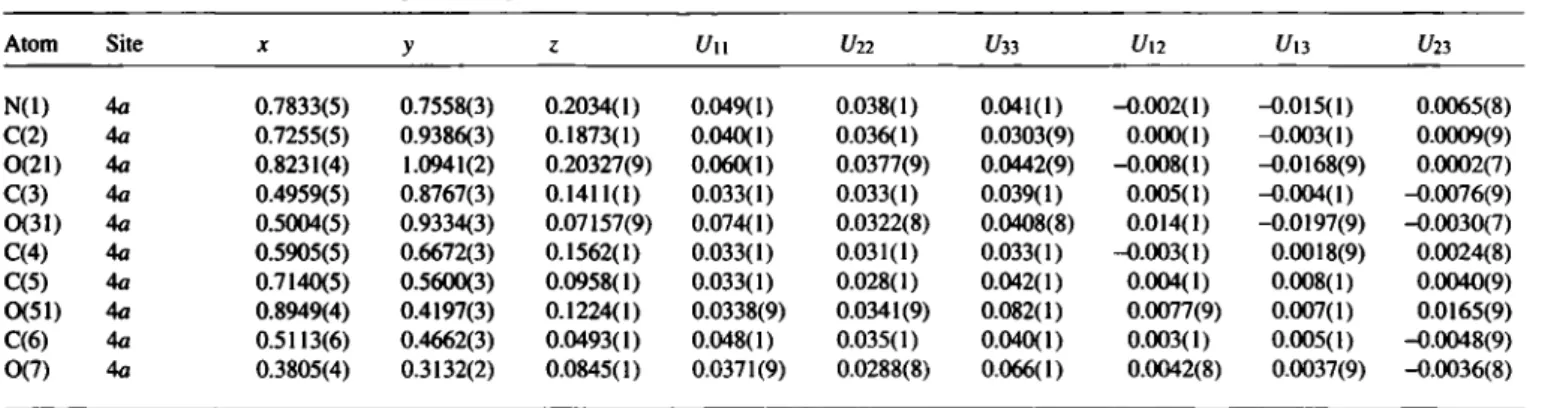
Table 2. Final atomic coordinates and displacement parameters (in Â2)
Source of material: The title compound (alternative name: 3- amino-3-deoxy-L-xylono-1,3-lactam; see réf. 1 ) was prepared by [2+2]-cycloaddition of (2/?)-2-0-benzyl-glyceraldehyde-A/-(4- methoxyphenyl)imine (see refs. 2,3) with acetoxyacetylchloride in CH2Cl2/Et3N at 243 Κ (see ref. 1), followed by oxidative N-dearylation, saponification of the ester group and hydrogeno- lytic cleavage of the benzyl ether groups. The azetidin-2-one was crystallized from methanol.
C5H9NO4, orthorhombic, P2i2i2i (No. 19), a =5.094(1) Â, b =6.894(2) Â, c =19.230(5) Â, V =675.4 Â
3, Ζ =4, R(F)=0.038, R^F
2) =0.084.
Table 3. Final atomic coordinates and displacement parameters (in Â2)
Atom Site
X y ζ U,so
H(l)
4a
0.898(6) 0.706(5) 0.230(2) 0.056(9) H(3)4a
0.333(5) 0.907(4) 0.163(1) 0.039(7) H(31)4a
0.451(7) 1.049(5) 0.073(2) 0.07(1) H(4)4a
0.463(5) 0.585(4) 0.179(1) 0.036(6) H(5)4a
0.826(6) 0.647(4) 0.070(1) 0.044(7) H(51)4a
0.823(7) 0.331(5) 0.143(2) 0.07(1) H(6A)4a
0.3840(6) 0.5627(3) 0.0348(1) 0.050 H(6B)4a
0.5964(6) 0.4156(3) 0.0080(1) 0.050 H(7)4a
0.230(7) 0.346(4) 0.097(2) 0.053(9)Atom Site U11 t/22 Í/33 U12 U13 C/23
N(l)
4a
0.7833(5) 0.7558(3) 0.2034(1) 0.049(1) 0.038(1) 0.041(1) -0.002(1) -0.015(1) 0.0065(8) C(2) 4a 0.7255(5) 0.9386(3) 0.1873(1) 0.040(1) 0.036(1) 0.0303(9) 0.000(1) -0.003(1) 0.0009(9) 0(21)4a
0.8231(4) 1.0941(2) 0.20327(9) 0.060(1) 0.0377(9) 0.0442(9) -0.008(1) -0.0168(9) 0.0002(7) C(3)4a
0.4959(5) 0.8767(3) 0.1411(1) 0.033(1) 0.033(1) 0.039(1) 0.005(1) -0.004(1) -0.0076(9) CK3I) 4a 0.5004(5) 0.9334(3) 0.07157(9) 0.074(1) 0.0322(8) 0.0408(8) 0.014(1) -0.0197(9) -0.0030(7) C(4) 4a 0.5905(5) 0.6672(3) 0.1562(1) 0.033(1) 0.031(1) 0.033(1) -0.003(1) 0.0018(9) 0.0024(8) C(5)4a
0.7140(5) 0.5600(3) 0.0958(1) 0.033(1) 0.028(1) 0.042(1) 0.004(1) 0.008(1) 0.0040(9) 0(51) 4a 0.8949(4) 0.4197(3) 0.1224(1) 0.0338(9) 0.0341(9) 0.082(1) 0.0077(9) 0.007(1) 0.0165(9) C(6)4a
0.5113(6) 0.4662(3) 0.0493(1) 0.048(1) 0.035(1) 0.040(1) 0.003(1) 0.005(1) -0.0048(9) 0(7)4a
0.3805(4) 0.3132(2) 0.0845(1) 0.0371(9) 0.0288(8) 0.066(1) 0.0042(8) 0.0037(9) -0.0036(8)Acknowledgment. We are grateful to the Fonds der Chemischen Industrie for financial support.
References
1. Krämer, Β. Franz, T.; Picasso, S.; Pruschke, P.; Jäger, V: Glycono-1,3- lactams, Xylo Series: Stereoselctive Access by [2+2]Cycloaddition, Exploratory Transformations, and Discovery of a New, Highly Selective Inhibitor of Glucoamylases. Synlett. In print.
2. Franz, T.; Hein, M.; Veith, U.; Jäger, V.; Peters, E. M.; Peters, K.; von Schnering, H.G.: Einfache und variable Synthese optisch aktiver 1,2-Ami- noalkohole durch Grignard-Reaktion an Λ/,Ο-Dibenzylglycerinaldimin und -lactaldimin. Angew. Chem. 106 (1994) 1308-1311; Angew. Chem.
Int. Ed. Engl. 33 (1994) 1305-1308.
3. Franz, T.: Stereoselektive Synthesen von 3-Amino-l,2-diolen, Isoxa- zolinen und ß-Lactamen aus 2-0-Benzylglycerinaldehyd - Schlüssel- strukturen zum Aufbau von Natur- und Wirkstoffen. Dissertation, University of Würzburg, Germany 1992.