Zeitschrift für Kristallographie - New Crystal Structures 213, 65-66 65
© by R. Oldenbourg Verlag, München 1998
Crystal structure of l,3:2,4-di-0-ethyIidene-L-xylose oxime, C9H15NO5
S. Henkel, T. Leibold and V. Jäger
Universität Stuttgan, Instituí für Organische Chemie, Pfaffenwaldring 55, D - 7 0 5 6 9 Stuttgart, Germany Received January 13, 1997. C S D - N o . 4 0 2 7 9 8
Table 1. Parameters used for the X-ray data collection
Source of material: The title compound (see ref. 1 and 2) was prepared by reaction of hydroxylamine (see ref. 3) with the corresponding aldehyde. The latter was obtained in a two-step sequence from L-sorbitol (see ref. 2).
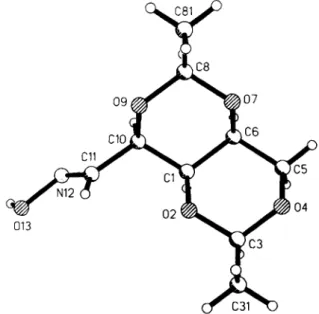
In the crystal the molecules are cross-linked by intermolecular hydrogen bridges of the oxime group (N-013B-H) to oxygen atoms 02A and 09A of the two dioxane rings of the neighbour molecule.
C9H15NO5, orthoriiombic, Р2\2\2\ (No. 19), a =8.783(1) Â,
¿7=9.622(1) À, с =12.811(2) A, V=1082.7Â^ Ζ =4, =0.056,
=0.125.
Table 3. Final atomic coordinates and displacement parameters (in λ^)
Crystal: colorless block, size 0.35 χ 0.4 χ 0.5 mm Wavelength: Mo Ka radiaüon (0.71073 A)
μ:
1.09 cm"'Difftactometer: Nicolet P3
Scan mode: Wyckoff
^nuasuremem'· 293 К
20п,ал: 56°
ЩНк[)тири·· 1452
Criterion for Io. /ο>2σ(/ο) 140
Programs: SHELXS-86, SHELXL-93
Table 2. Fmal atomic coordinates and displacement parameters (in Â^)
Atom Site χ У ζ ί/ύο
H(l) 4a 0.1438(4) 0.4577(3) 0.1921(3) 0.049 H(3) 4<z 0.0767(4) 0.6077(4) 0.0480(2) 0.050 H(31A) 4a 0.2600(5) 0.7632(4) -0.0116(3) 0.091 H(31B) Λα 0.1974(5) 0.8778(4) 0.0641(3) 0.091 H(31C) 4a 0.0973(5) 0.8259(4) -0.0295(3) 0.091 H(5A) Aa -0.1336(4) 0.5643(4) 0.1587(3) 0.055 H(5B) Λα -0.1867(4) 0.6810(4) 0.2362(3) 0.055 H(6) Λα -0.0364(4) 0.5050(3) 0.3190(3) 0.054 H(8) Λα 0.1043(4) 0.5697(5) 0.4710(3) 0.068 H(81A) Λα 0.2308(6) 0.7445(7) 0.5644(3) 0.126 H(81B) Λα 0.0626(6) 0.7908(7) 0.5430(3) 0.126 H(81C) Λα 0.1956(6) 0.8516(7) 0.4754(3) 0.126 H(10) Λα 0.2544(4) 0.4286(3) 0.3527(3) 0.056 H(ll) Λα 0.4842(4) 0.5788(4) 0.2417(3) 0.060 H(13) Λα 0.668(9) 0.315(8) 0.240(6) 0.17(3)
Atom Site χ У ζ Un U22 U33
t/12
Un £/23C d ) Λα 0.1598(4) 0.5422(3) 0.2333(3) 0.037(2) 0.029(1) 0.057(2) 0.000(1) -0.002(2) 0.000(2) 0(2) Λα 0.2146(2) 0.6506(2) 0.1665(2) 0,034(1) 0.037(1) 0.042(1) -0.001(1) 0.0023(9) -0.001(1) C(3) Λα 0.1040(4) 0.6889(4) 0.0902(2) 0.046(2) 0.043(2) 0.037(2) 0.003(2) -0.001(1) -0.008(1) C(3I) Λα 0.1706(5) 0.7987(4) 0.0223(3) 0.061(2) 0.075(3) 0.046(2) 0.008(2) 0.018(2) 0.014(2) 0(4) Λα -0.0262(3) 0.7424(2) 0.1389(2) 0.036(1) 0.043(1) 0.042(1) 0.002(1) -0.002(1) 0.005(1) C(5) Λα -0.0986(4) 0.6403(4) 0.2023(3) 0.031(1) 0.048(2) 0.058(2) -0.006(1) -0.001(2) 0.010(2) C(6) Λα 0.0094(4) 0.5852(3) 0.2839(3) 0.033(2) 0.045(2) 0.057(2) -0.004(1) 0.002(2) 0.015(2) 0(7) Λα 0.0352(3) 0.6936(3) 0.3578(2) 0.039(1) 0.064(2) 0.041(1) 0.008(1) 0.003(1) 0.004(1) C(8) Λα 0.1409(4) 0.6530(5) 0.4348(3) 0.047(2) 0.081(3) 0.041(2) 0.005(2) 0.001(2) 0.014(2) C(81) Λα 0.1591(6) 0.7706(7) 0.5113(3) 0.076(3) 0.130(5) 0.046(2) 0.025(4) -0.007(2) -0.020(3) 0(9) Λα 0.2852(3) 0.6267(3) 0.3892(2) 0.039(1) 0.054(1) 0.054(1) 0.005(1) -0.004(1) 0.001(1) C(10) Λα 0.2799(4) 0.5147(3) 0.3157(3) 0.039(2) 0.036(2) 0.065(2) 0.001(2) 0.000(2) 0.010(2) C ( l l ) Λα 0.4370(4) 0.5024<4) 0.2721(3) 0.037(2) 0.039(2) 0.075(2) 0.004(1) -0.002(2) 0.007(2) N(12) Λα 0.5041(4) 0.3878(3) 0.2770(4) 0.044(2) 0.041(2) 0.125(3) 0.006(1) 0.013(2) 0.011(2) 0(13) Λα 0.6486(4) 0.3978(3) 0.2315(4) 0.052(2) 0.049(2) 0.164(4) 0.008(1) 0.030(2) 0.012(2)
66
l , 3 : 2 , 4 - D i - 0 - e t h y l i d e n e - L - x y l o s e o x i m eAcknowledgments. We are grateful to the Fonds der Chemischen Industrie, Bayer AG and Hoechst AG for financial support. We thank Mr. Rolf Kugler, participant of the undergraduate research programme, Universität Stuttgart 1994, for experimental assistance and Dr. Wolfgang Frey for help with the preparation of the Tiles.
References
I. Leibold, T.: Furan-Nitriloxid-Cycloaddukte - diastereomerenreine Vor- läufer für die Synthese von biologisch aktiven Desoxynojirimycin- und Castanospermin-Analoga. Dissertation, Universität Stuttgart. Germany 1995.
Hockett, R. C.; Schaefer. F. C.: Lead tetraacetate oxidations in the sugar group. X. Crystalline 2,4,3,5-diethylidene-aldehydo-L-xylose and several of its derivatives. J. Am. Chem. Soc. 69 ( 1947) 849-851.
Hoffmann, R. W.; Eichler, G.: Endesfelder. Α.: Addition von Allylboron- säureestem an Schiffsche Basen und Oxime. Liebigs Ann. Chem. (1983) 2000-2007.
Sheldrick, G. M,: Phase Annealing in SHELX-90: Direct Methods for Large Structures. Acta Crystallogr. A46 (1990) 467-473.
Sheldrick, G. M.: SHELXL-93. Program for refining crystal structures.
University of Göttingen. Germany 1993.