Crystal structure of (3R,4R,5R,6R)-2-benzyl-3-methyl-4,5-O- isopropylidenedioxy-6-(N-benzylhydroxylamino)-tetrahydro- 1,2-oxazine, C 22 H 28 N 2 O 4
Wolfgang Frey, Hua Yang, Andreas Marc Palmer and Volker Jäger
*Universität Stuttgart, Institut für Organische Chemie, Pfaffenwaldring 55, 70569 Stuttgart, Germany Received June 9, 2010, accepted and available on-line July 23, 2010; CCDC no. 1267/3111
Abstract
C
22H
28N
2O
4, orthorhombic, P2
12
12
1(no. 19), a = 10.213(2) Å, b = 13.211(2) Å, c = 15.574(3) Å, V = 2101.3 Å
3, Z = 4, R
gt(F) = 0.042, wR
ref(F
2) = 0.105, T = 293 K.
Source of material
The title compound was obtained by reaction of (Z)-4,5-dideoxy- 2,3-O-isopropylidene-D-erythro-4-pentenose N-benzyl nitrone with N-benzylhydroxylamine in dry dichloromethane in the pres- ence of magnesium sulfate [1,2,3]. The resulting colourless solid was purified by recrystallization from petroleum ether/ethyl ace- tate, m.p. 154 °C (cf. m.p. 154 °C [2]), [ ] *
D20= –91 (c = 1.10, CH
2Cl
2), cf. [ ] *
D20= –94 (c = 1.07, CH
2Cl
2) [1,2].
Experimental details
H atoms were located on difference Fourier map, but refined with fixed individual displacement parameters using a riding model with d(C—H) = 0.93 - 0.98 Å, excluding the hydrogen of the hydroxy group which was refined free, because of its relevance in hydrogen bond interactions. In addition, the methyl groups were allowed to rotate but not to tip. The measured range of h k l is pos- sibly not suitable for characterisation of the absolute structure.
Discussion
The title compound crystallizes with one independent molecule in the asymmetric unit (figure, top). The oxazine ring shows a chair conformation and the dioxolane moiety has an envelope conformation where C2 is out of plane. The hydroxy function O2–H2A establishes a bifurcated intramolecular hydrogen bond where N1 and O1 of the oxazine ring act as acceptors. The H2A···O1 distance is 2.60(2) Å and the angle O2–H2A···O1 is 109(1)°. The H2A···N1 distance is 2.11(2) Å with an angle O2–H2A···N1 of 140(2)° which characterizes the much stronger part of this bifurcated interaction. A weak, intramolecular elec- trostatic interaction is observed between the methylene group of the benzyl moiety attached to the hydroxyamino function as do- nor and the ,-system of the benzyl group attached on N1 of the oxazine ring as acceptor. The H16A···X1A (center of the ,-sys- tem) distance is 2.86 Å and the angle C16–H16A···X1A is 166°.
Finally there is also evidence of a weak intermolecular electro- static contact between C13–H13 of a phenyl ring as donor and O1 of the oxazine moiety as acceptor. The H13···O1 distance is 2.50 Å with an angle C13–H13···O1 of 144°. The view along [100] of the unit cell (figure, bottom) shows a layer-type packing organization of the molecules. There is an alternate stacking in two directions ([010] and [001]) between the oxazine core and the benzyl moieties on the other hand.
Z. Kristallogr. NCS 225 (2010) 587-588 / DOI 10.1524/ncrs.2010.0256 587
© by Oldenbourg Wissenschaftsverlag, München
_____________
* Correspondence author (e-mail: jager.ioc@oc.uni-stuttgart.de)
588 C22H28N2O4
O(1) 4a 0.18785(8) 0.38365(6) 0.14372(5) 0.0429(4) 0.0311(3) 0.0333(3) 0.0014(3) 0.0014(3) 0.0000(3) N(1) 4a 0.12754(9) 0.37239(7) 0.22920(6) 0.0363(4) 0.0330(4) 0.0340(4) −0.0034(3) −0.0023(3) 0.0043(3) C(1) 4a 0.1990(1) 0.48878(8) 0.12226(7) 0.0403(5) 0.0335(4) 0.0348(4) −0.0022(4) −0.0015(4) 0.0054(4) N(2) 4a 0.0784(1) 0.54082(7) 0.10271(6) 0.0461(5) 0.0353(4) 0.0391(4) 0.0025(4) −0.0072(4) 0.0036(4) O(2) 4a 0.0019(1) 0.55421(8) 0.17991(6) 0.0571(5) 0.0518(5) 0.0463(5) 0.0192(4) −0.0034(4) −0.0058(4) C(2) 4a 0.2779(1) 0.54567(8) 0.19004(7) 0.0395(5) 0.0320(4) 0.0425(5) −0.0046(4) −0.0040(4) 0.0047(4) O(3) 4a 0.39237(9) 0.52838(8) 0.31786(6) 0.0397(4) 0.0592(5) 0.0440(4) −0.0114(4) −0.0070(3) −0.0022(4) C(3) 4a 0.2658(1) 0.50800(8) 0.28204(7) 0.0344(4) 0.0373(5) 0.0370(5) −0.0004(4) −0.0026(4) −0.0033(4) O(4) 4a 0.41361(9) 0.52963(8) 0.17183(6) 0.0386(4) 0.0590(5) 0.0452(4) −0.0106(4) 0.0008(3) 0.0062(4) C(4) 4a 0.2353(1) 0.39481(8) 0.28989(7) 0.0350(5) 0.0380(5) 0.0350(4) −0.0025(4) −0.0036(4) 0.0047(4) C(5) 4a 0.1967(2) 0.3700(2) 0.38199(9) 0.0661(9) 0.082(1) 0.0382(6) −0.0228(8) −0.0061(6) 0.0150(6) C(6) 4a 0.4834(1) 0.5476(1) 0.24967(9) 0.0379(5) 0.0484(6) 0.0513(6) −0.0069(5) −0.0039(5) 0.0028(5) C(7) 4a 0.5972(2) 0.4744(2) 0.2545(1) 0.0530(8) 0.087(1) 0.073(1) 0.0175(8) −0.0006(8) 0.0070(9) C(8) 4a 0.5270(2) 0.6574(1) 0.2545(1) 0.065(1) 0.0612(9) 0.091(1) −0.0277(8) −0.0083(9) 0.0015(9) C(9) 4a 0.0894(1) 0.26459(9) 0.23094(9) 0.0490(6) 0.0342(5) 0.0566(7) −0.0083(4) −0.0122(6) 0.0113(5) C(10) 4a −0.0148(1) 0.24234(8) 0.16490(8) 0.0461(6) 0.0317(4) 0.0481(6) −0.0086(4) −0.0049(5) 0.0045(4) C(11) 4a 0.0145(2) 0.1877(1) 0.0914(1) 0.0548(8) 0.0517(7) 0.0660(9) −0.0080(6) 0.0045(7) −0.0100(7) C(12) 4a −0.0809(2) 0.1705(1) 0.0293(1) 0.081(1) 0.0668(9) 0.0586(9) −0.0183(9) 0.0012(8) −0.0181(7) C(13) 4a −0.2059(2) 0.2068(1) 0.0406(1) 0.073(1) 0.0582(8) 0.0578(8) −0.0183(7) −0.0199(8) −0.0007(6) C(14) 4a −0.2364(2) 0.2595(1) 0.1141(1) 0.0470(7) 0.0512(7) 0.0696(9) −0.0074(6) −0.0118(7) 0.0044(6) C(15) 4a −0.1413(1) 0.2771(1) 0.17633(9) 0.0495(6) 0.0432(6) 0.0498(6) −0.0050(5) −0.0033(5) −0.0008(5) C(16) 4a −0.0011(1) 0.48879(9) 0.03790(8) 0.0483(6) 0.0382(5) 0.0418(5) 0.0024(5) −0.0093(5) 0.0025(4) C(17) 4a −0.1100(1) 0.5567(1) 0.00618(8) 0.0466(6) 0.0461(6) 0.0377(5) 0.0038(5) −0.0082(4) 0.0024(4) C(18) 4a −0.2394(2) 0.5303(2) 0.0144(2) 0.0529(8) 0.083(1) 0.102(1) −0.0071(8) −0.0178(9) 0.030(1) C(19) 4a −0.3380(2) 0.5943(2) −0.0158(2) 0.0496(9) 0.128(2) 0.131(2) 0.009(1) −0.023(1) 0.027(2) C(20) 4a −0.3067(2) 0.6836(2) −0.0561(1) 0.077(1) 0.091(1) 0.072(1) 0.037(1) −0.025(1) −0.000(1) C(21) 4a −0.1788(2) 0.7107(1) −0.0634(1) 0.092(1) 0.0573(8) 0.0515(8) 0.0248(9) −0.0108(8) 0.0069(6) C(22) 4a −0.0801(2) 0.6484(1) −0.0326(1) 0.0607(8) 0.0484(7) 0.0523(7) 0.0064(6) −0.0040(6) 0.0083(6) Table 3. Atomic coordinates and displacement parameters (in Å2).
Atom Site x y z U11 U22 U33 U12 U13 U23
H(1) 4a 0.2515 0.4914 0.0696 0.043
H(2A) 4a 0.002(2) 0.497(1) 0.206(1) 0.059(5)
H(2) 4a 0.2576 0.6181 0.1874 0.046
H(3) 4a 0.1991 0.5474 0.3125 0.044
H(4) 4a 0.3130 0.3554 0.2740 0.043
H(5A) 4a 0.1917 0.2979 0.3889 0.093
Table 2. Atomic coordinates and displacement parameters (in Å2).
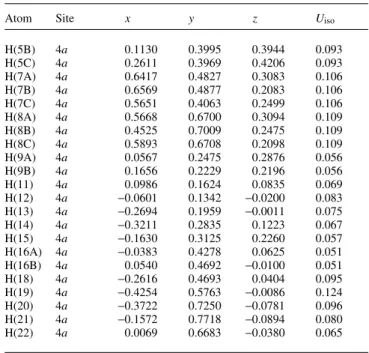
Atom Site x y z Uiso
Crystal: colorless prism, size 0.5 × 0.6 × 0.9 mm Wavelength: Mo K*radiation (0.71073 Å)
-: 0.84 cm−1
Diffractometer, scan mode: Nicolet P3, Wyckoff
2+max: 65°
N(hkl)measured, N(hkl)unique: 8223, 7601 Criterion for Iobs, N(hkl)gt: Iobs> 2((Iobs), 6572 N(param)refined: 261
Programs: SHELXS-97, SHELXL-97,
SHELXTL-Plus [4]
Table 1. Data collection and handling.
H(5B) 4a 0.1130 0.3995 0.3944 0.093
H(5C) 4a 0.2611 0.3969 0.4206 0.093
H(7A) 4a 0.6417 0.4827 0.3083 0.106
H(7B) 4a 0.6569 0.4877 0.2083 0.106
H(7C) 4a 0.5651 0.4063 0.2499 0.106
H(8A) 4a 0.5668 0.6700 0.3094 0.109
H(8B) 4a 0.4525 0.7009 0.2475 0.109
H(8C) 4a 0.5893 0.6708 0.2098 0.109
H(9A) 4a 0.0567 0.2475 0.2876 0.056
H(9B) 4a 0.1656 0.2229 0.2196 0.056
H(11) 4a 0.0986 0.1624 0.0835 0.069
H(12) 4a −0.0601 0.1342 −0.0200 0.083
H(13) 4a −0.2694 0.1959 −0.0011 0.075
H(14) 4a −0.3211 0.2835 0.1223 0.067
H(15) 4a −0.1630 0.3125 0.2260 0.057
H(16A) 4a −0.0383 0.4278 0.0625 0.051
H(16B) 4a 0.0540 0.4692 −0.0100 0.051
H(18) 4a −0.2616 0.4693 0.0404 0.095
H(19) 4a −0.4254 0.5763 −0.0086 0.124
H(20) 4a −0.3722 0.7250 −0.0781 0.096
H(21) 4a −0.1572 0.7718 −0.0894 0.080
H(22) 4a 0.0069 0.6683 −0.0380 0.065
Table 2. Continued.
Atom Site x y z Uiso
Acknowledgments. We are grateful to Fonds der Chemischen Industrie/
BWFT der Bundesrepublik Deutschland for a doctoral stipend (to A. M.
Palmer) and to Prof. Dr. J. Senn-Bilfinger, Altana Pharma (now Nycomed), Konstanz, for financial support.
References
1. Palmer, A. M.: Die Cope-House-Cyclisierung ungesättigter Hydroxylamine - ein neuer Weg zu Strukturen mit Glykosidase- Hemmaktivität. Dissertation, Universität Stuttgart, 2001.
2. Yang, H.: Nucleophilic Additions to&,'-Unsaturated Nitrones: An Ap- proach to Functionalized Pyrrolidines and Tetrahydro-1,2-oxazines. Syn- thesis of 3-Phenylisothreonines: Analogues of the C-13 Side-Chain of Taxotere by Means of Chiral Borane Reagents. Dissertation, Universität Stuttgart, 2010.
3. Jäger, V.; Bierer, L.; Dong, H.-Q.; Palmer, A. M.; Shaw, D.; Frey, W.:
New Heterocyclic Structures from Unsaturated Aldehyde Derivatives. In- hibition of*-Fucosidases. J. Heterocycl. Chem. 37 (2000) 455-465.
4. Sheldrick, G. M.: A short history of SHELX. Acta Crystallogr. A64 (2008) 112-122.