Original article:
URINARY
PCA3
DETECTION IN PROSTATE CANCER BY MAGNETIC NANOPARTICLES COUPLED WITH COLORIMETRICENZYME-LINKED OLIGONUCLEOTIDE ASSAY
Vichanan Yamkamon1, Khin Phyu Pyar Htoo2,3, Sakda Yainoy2, Thummaruk Suksrichavalit4, Tienrat Tangchaikeeree5, Warawan Eiamphungporn2*
1 Department of Clinical Microscopy, Faculty of Medical Technology, Mahidol University, Bangkok, Thailand
2 Department of Clinical Microbiology and Applied Technology, Faculty of Medical Technology, Mahidol University, Bangkok, Thailand
3 Department of Medical Laboratory Technology, University of Medical Technology, Mandalay, Myanmar
4 Center of Data Mining and Biomedical Informatics, Faculty of Medical Technology, Mahidol University, Bangkok, Thailand
5 Center for Research and Innovation, Faculty of Medical Technology, Mahidol University, Bangkok, Thailand
* Corresponding author: Department of Clinical Microbiology and Applied Technology, Faculty of Medical Technology, Mahidol University, Bangkok, Thailand, Telephone:
+66 2 441 4371 ext. 2628, Fax: +66 2 441 4380, E-mail: warawan.eia@mahidol.ac.th
http://dx.doi.org/10.17179/excli2020-1036
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0/).
ABSTRACT
PCA3 is one of the most prostate cancer-specific genes described to date. Of note, PCA3 expression is detectable at high level in the urine of prostate cancer (PCa) patients. Accordingly, PCA3 is an ideal biomarker for PCa diagnosis. Several techniques for the measurement of this biomarker in urine have been developed but there are still some drawbacks. In this study, magnetic nanoparticle-based PCR coupled with streptavidin-horseradish per- oxidase and a substrate for colorimetric detection was established as a potential assay for urinary PCA3 detection.
The method provided a high specificity for PCA3 gene in LNCaP prostate cancer cell line. Additionally, this technique could detect PCA3 at femtogram level which was approximately 1,000-fold more sensitive than the conventional RT-PCR followed by agarose gel electrophoresis. The effectiveness of the method was assessed by PCA3 detection in clinical specimens. The relative PCA3 expression of PCa patients determined by this assay was significantly greater than that of benign prostatic hyperplasia (BPH) patients and healthy controls. The results of our test were comparable with the results of qRT-PCR. The proposed method is promising to distinguish between cancerous and non-cancerous groups. Altogether, this simple assay is practicable and useful for prostate cancer diagnosis.
Keywords: PCA3, prostate cancer, urine, magnetic nanoparticle, colorimetric assay
INTRODUCTION
Prostate cancer (PCa) is the most common type of cancer diagnosed in men and a leading cause of cancer-related deaths (Ma and Huang, 2017). The incidence of prostate can- cer has been reported in speedily growing trend worldwide (Siegel et al., 2018; Taitt, 2018). In clinical practice, prostate specific antigen (PSA) blood test combined with digi- tal rectal examination (DRE) is currently used for PCa screening and monitoring (Smith et al., 2004). Based on suspicious DRE and/or increased PSA level in serum, biopsy which is more invasive is manipulated (Catalona et al., 1994). A serum PSA level of below 4 ng/mL is considered normal and it increases significantly in men with prostate cancer.
Nevertheless, this biomarker is specific to tis- sue of prostate origin but not only in PCa, PSA level is often elevated in benign prostatic hyperplasia (BPH) and prostatitis (Roobol et al., 2007). Moreover, previous studies indi- cated 15 % of PCa patients have PSA level below 4.0 ng/mL which leads to false nega- tive results (Thompson et al., 2004). As a re- sult of limited specificity of PSA, role of PSA for PCa diagnosis still remains controversial (Cucchiara et al., 2018; Catalona, 2018). Cur- rently, the United States Preventive Services Task Force (USPSTF) has new guidelines that PSA test may be less beneficial for some men and it should not be screened routinely (US Preventive Services Task Force et al., 2018).
Therefore, the new finding of more specific PCa markers for screening and diagnosis is essential.
Prostate cancer antigen 3 (PCA3), also known as differential display code 3 or DD3, is a non-coding RNA and it is overexpressed in 95 % of primary PCa tissue specimen (Bussemakers et al., 1999). However, it was undetectable in bladder, breast, cervix, endo- metrium, kidney, ovary or testis tumors (Loeb and Partin, 2011). Notably, the expression of PCA3 in BPH and normal tissues was very low (Wang et al., 2014). Interestingly, PCA3 can be detected in post DRE urine samples, thus it was proposed as a promising bi-
omarker for PCa. The US Food and Drug Ad- ministration (FDA) recently approved Pro- gensa PCA3 assay as the first urine-based mo- lecular test for clinical diagnosis recom- mended for men with elevated serum PSA and a previous negative biopsy (Cui et al., 2016). PCA3 assay is commercially available, but it is expensive and time-consuming as well as it needs a complicated instrument. To overcome these limitations, a simple, sensi- tive and cost-effective assay for PCA3 detec- tion in urine samples is absolutely crucial.
In the last two decades, nanotechnology and nanoscience have been greatly employed in various fields, such as molecular diagnos- tics, biomedicine, bioseparation and drug de- livery (Akbarzadeh et al., 2012; Mody et al., 2014). Particularly, nanoparticles (NPs) have been used as signal amplification tools due to their small size (1-100 nm), corresponding high surface-to-volume ratio and unusual tar- get binding properties (Perez et al., 2011).
Among the broad spectrum of nanoparticles, magnetic nanoparticles (MNPs) are a major class of nano-scale material currently under extensive development for a wide range of ap- plications (Shabestari Khiabani et al., 2017).
MNPs have been exploited because of their exclusive physical properties, magnetic sus- ceptibility, biocompatibility, stability, easy functionalization and many more relevant features (Amiri et al., 2019). MNPs function- alized with targeting moieties enable the effi- cient collection and separation of target mol- ecules in a simple and rapid process without any filtration or centrifugation (Kim and Sear- son, 2017). Recently, MNP-based polymerase chain reaction (PCR) coupled with colorimet- ric assay has been developed for gene detec- tion of several diseases (Jangpatarapongsa et al., 2011; Tangchaikeeree et al., 2017a). This strategy combines the versatility of colorimet- ric assay based on enzyme-substrate system and the exponential amplification capacity of PCR using MNPs functionalized with oligo- nucleotide primer. The PCR method is a highly sensitive and specific technique that can amplify a huge number of DNA copies
from a single target DNA, allowing an effec- tive detection from a small amount of sample.
Magnetic field has been applied for isolation of nucleic acid after amplification to increase the sensitivity of the test. Colorimetric assay is a simple and sensitive method that can be visually detected or requires only a spectro- photometer. Taken together, this methodol- ogy is facile, highly specific and provides bet- ter sensitivity when compared with the con- ventional RT-PCR followed by agarose gel electrophoresis.
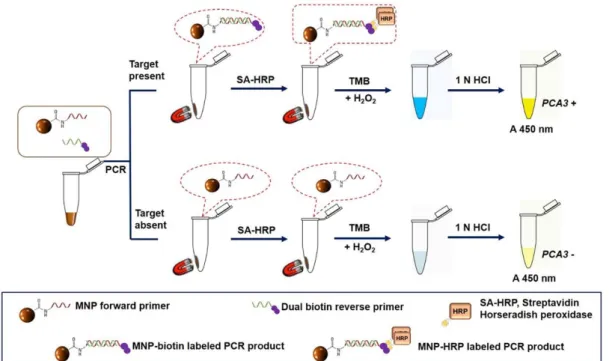
In this study, the MNP-based PCR cou- pled with colorimetric enzyme-linked oligo- nucleotide assay was developed to detect uri- nary PCA3 for PCa determination. This tech- nique based on RT-PCR using MNP-labeled forward primer and dual biotin-labeled re- verse primer combined with horseradish pe- roxidase-streptavidin detection system. The schematic diagram of the assay is illustrated in Figure 1. The proposed method showed high level of analytical sensitivity as well as specificity and it could potentially be used for the detection of PCA3. Moreover, this meth- odology could differentiate PCa patients from both healthy controls and BPH patients based on PCA3 level in urine. In conclusion, our as-
say holds a promising prospect for PCa diag- nosis.
MATERIALS AND METHODS Materials and reagents
MNPs were synthesized by co-precipita- tion of FeCl3 and FeCl2, then they were car- boxylate-functionalized by emulsion poly- merization according to previously described procedures (Montagne et al., 2002; Braconnot et al., 2013). For MNP immobilization, all specific primers including amino (NH2)- labeled forward and dual biotin-labeled re- verse primers were ordered from Integrated DNA Technologies, Inc. (Skokie, IL, USA).
All chemicals required for the immobilization were from Sigma Aldrich (St. Louis, MO, USA). SsoAdvanced Universal SYBR Green Supermix was purchased from Bio-Rad (Hercules, CA, USA). RevertAid First Strand cDNA synthesis kit and Phusion high-fidelity DNA polymerase were obtained from Thermo Fisher Scientific (Waltham, MA, USA). Media and reagents used for cell cul- ture were purchased from Hyclone (Logan, UT, USA) and Gibco (Carlsbad, CA, USA).
Figure 1: A schematic illustration of the MNP-based PCR coupled with colorimetric enzyme-linked oli- gonucleotide assay for PCA3 detection in urine sediments
Cell lines
LNCaP clone FGC (ATCC CRL-1740) prostate cancer cell line was used as a positive control for PCA3. RWPE-1 (ATCC CRL- 11609) prostate epithelial cell line, MDA- MB-436 (ATCC HTB-130) breast cancer cell line and K562 (ATCC CCL-243) chronic my- elogenous leukemia cell line were used as negative controls for PCA3. All cell lines were purchased from the American Type Cul- ture Collection (ATCC, Rockville, MD, USA). They were maintained in defined me- dia and cultured under the conditions as pre- viously described (Htoo et al., 2019; Promkan et al., 2013; Suangtamai and Tanyong, 2016).
Urine sample collection and preparation Spot urine samples were collected from healthy male subjects (n = 5) and first voided post-DRE urine samples were received from BPH patients (n = 5) as well as from PCa pa- tients (n = 5). All patients were diagnosed based on histopathological analysis after prostate biopsy. Samples were provided by the Division of Urology, Department of Sur- gery, Faculty of Medicine, Ramathibodi Hos- pital, Mahidol University. The present study was approved by the Committee on Human Rights Related to Research Involving Human Subjects, Faculty of Medicine, Ramathibodi
Hospital, Mahidol University (MURA2016/34). Written informed consent
was obtained from each individual in the study. After collection, urine samples were centrifuged at 4 °C, 3,000 rpm for 10 min.
The supernatants were discarded and cell pel- lets were washed twice by PBS, pH 7.0. Sub- sequently, TRIzol reagent (Invitrogen, CA, USA) was added to the sediments which were then stored at -20 °C until RNA isolation.
RNA extraction and cDNA synthesis
Total RNA was isolated from cell lines or from cell sediments of urine with TRIzol rea- gent. 500 ng of total RNA was converted to cDNA using RevertAid First Strand cDNA synthesis kit following the manufacturer’s in- structions and kept at -20 °C until use.
Immobilization of forward primer on MNPs Forward primer was immobilized on MNPs using carbodiimide crosslinking method as previously described (Nakajima and Ikada, 1995). Briefly, 1 mg of carboxy- lated MNPs was washed twice with 100 μL of 25 mM 2-(N-morpholino) ethanesulfonic acid (MES) buffer, pH 6.0. The washed MNPs were resuspended in the 50 μL of MES buffer and thoroughly mixed with 5 nmol of NH2- modified PCA3 forward primer. The mixture was incubated at room temperature with gen- tle shaking for 30 min. The surface of MNPs was subsequently activated by adding a freshly prepared N-(3-Dimethylaminopro- pyl)-N′-ethylcarbodiimide (EDC) solution (10 mg/mL) with the ratio of 1:1 (v/v). The activated MNPs were then incubated with shaking at 900 rpm, room temperature for overnight. Using external magnetic field, MNP-immobilized PCA3 forward primer was separated and supernatant was simultaneous- ly collected. The residual primer concentra- tion in the supernatant was measured at 260 nm using a NanoDrop2000c UV-Vis spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). Afterwards, the MNP- labeled forward primer was washed twice and completely resuspended in 50 μL of TE buffer (10 mM Tris–HCl, 1 mM EDTA, pH 8.0).
The MNP-labeled primer was stored at 4 °C until use.
Conventional RT-PCR
PCA3 amplification was performed using conventional primers. The sequences of pri- mers are shown in Table 1. PCR was carried out in a 50 µl reaction mixture containing 0.05 µg of cDNA template, 0.5 µM of each primer, 1x HF reaction buffer, 0.2 mM dNTPs and 0.4 U Phusion high-fidelity DNA poly- merase. The cycling procedures were per- formed as followed: initial denaturation at 98 °C for 30 sec followed by 30 cycles of de- naturation at 98 °C for 10 s, annealing at 60 °C for 30 s, extension at 72 °C for 30 s and a final extension step at 72 °C for 5 min. Of note, a housekeeping gene, GADPH gene was
used as a control to evaluate the cDNA qual- ity. To amplify GADPH, the cycling proce- dures were set for 5 min at 95 °C, followed by 30 cycles of 30 s at 95 °C, 30 s at 50 °C, and 30 s at 72 °C, and finally extension 5 min at 72 °C. After amplification, 10 µL PCR prod- ucts were analyzed by gel electrophoresis.
The PCR amplicon sizes of PCA3 and GADPH were 167 bp and 106 bp, respec- tively.
Optimization of primer concentration for MNP-based PCR
Prior to perform the MNP-based PCR, the concentrations of MNP-labeled forward pri- mer were optimized. In brief, PCR was achieved in a 50 µL reaction mixture contain- ing 0.05 µg of cDNA template, 0.5 µM re- verse primer, 1× HF reaction buffer, 0.2 mM dNTPs and 0.4 U Phusion high-fidelity DNA polymerase. Each concentration of MNP la- beled-forward primer (2, 4 and 6 µg) was in- dividually added to the PCR reaction. The MNP-based PCR amplification was per- formed with identical conditions of the con- ventional RT-PCR, as above mentioned.
MNP-based PCR coupled with colorimetric assay for PCA3 detection
The optimized procedure of MNP-based PCR for PCA3 detection was performed. The 6 μg MNP-labeled forward primer was ap- plied in the PCR reaction. The PCR was per- formed with identical conditions of the con- ventional RT-PCR as previously mentioned.
Subsequently, PCR products were detected by colorimetric method. Briefly, the MNP-
bound PCR products were washed with de- ionized water using magnetic separation. Af- ter washing, the MNP-bound PCR products were mixed with 50 μL of horseradish perox- idase-conjugated streptavidin (SA-HRP) in 0.1 % BSA and incubated for 30 min in a dark chamber at room temperature. The SA-HRP conjugated PCR products were washed with 100 μL of 0.1 % BSA and then with deionized water. The conjugated PCR products were in- cubated with 50 μL of SureBlueTM TMB (3, 3', 5, 5'-tetramethylbenzidine) microwell pe- roxidase substrate (KPL, Gaithersburg, MO, USA) for 10 min in the dark at room temper- ature. The reaction was stopped by 50 μL of 1 N HCl. The optical density (OD) at 450 nm was measured using microplate reader (BioTek Inc., Winooski, USA). The results were calculated to the relative OD450 which means the OD450 ratio of the sample to the non-template control (NTC).
Sensitivity and specificity tests
The analytical sensitivity of the assay was investigated. Serial ten-fold dilutions of cDNA from LNCaP cells ranging from 1 µg to 1 fg were prepared. Each dilution was em- ployed as a template for MNP-based PCR. To examine the analytical specificity, the cDNA samples from LNCaP, RWPE-1, MDA-MB- 436 and K562 cell lines were utilized as tem- plates for MNP-based PCR. The PCR product of NTC was used a negative control. The MNP-based PCR products were determined using an enzyme-substrate system in the same manner as described above.
Table 1: List of primers
Primers Sequence (5' 3')
PCA3 forward primer GCTGACTTTACCATCTGAGG
Amino-labeled forward primer (NH2-C6)-GCTGACTTTACCATCTGAGG
PCA3 reverse primer GATCTCTGTGCTTCCTTTTG
Dual biotin-labeled reverse primer (biotin)-GATCTCTGTGCTTCCTTTTG
GAPDH forward primer CAGCCTCAAGATCATCAGCA
GAPDH reverse primer TGTGGTCATGAGTCCTTCCA
qRT-PCR
qRT-PCR was achieved as a standard method to determine the expression of target genes. qRT-PCR primers are shown in Table 1. Each PCR reaction was performed in a 20 μL reaction mixture comprising 1×
SsoAdvanced Universal SYBR Green Super- mix, 0.5 μM of each primer and 0.025 µg of cDNA template. The qRT-PCR program for PCA3 and GAPDH was set for 3 min at 95 °C, followed by 40 cycles of 20 s at 95 °C and 30 s at 60 °C. The baseline threshold was ad- justed and the Ct was determined. Melt curve analysis was analyzed by CFX™ manager software 3.1 (Bio-Rad, Hercules, CA, USA).
Gel electrophoresis was conducted to confirm the presence of PCR products. The relative expression level of PCA3 was calculated by a 2-ΔΔCt relative quantification method accord- ing to the manufacturer's instruction.
Statistical analysis
Data were analyzed by SPSS PASW Sta- tistics 25 (SPSS Inc., Chicago, USA) and GraphPad Prism (version 7.03, GraphPad Software). They were represented as mean ± standard deviation (SD). The differences be- tween groups in each experiment (samples from cell lines and subjects) were compared using one-way ANOVA. Post hoc analysis was applied by using Dunnett’s t multiple comparison for testing of sensitivity and spec- ificity. Linear regression method was used to examine the relationship between the differ- ent concentrations of the standards and rela- tive absorbance values. Statistical signifi- cance was defined as a P-value < 0.05.
RESULTS Primer immobilized efficiency
To obtain MNP-labeled forward primer, 5 nmol NH2-modified PCA3 forward primer was immobilized onto the surface of 1 mg car- boxylated MNPs using EDC as a coupling agent. The residual PCA3 forward primer concentration in the supernatant after immo- bilization was measured to evaluate the bind- ing efficiency. The result demonstrated that the average concentration of the residual
PCA3 forward primer was 2.4 ± 0.02 nmol/
mg corresponding to the immobilized NH2- modified PCA3 forward primer at 2.6 ± 0.02 nmol/mg. The binding efficiency was rela- tively calculated to 51.9 %.
Optimization of primer concentration for MNP-based PCR
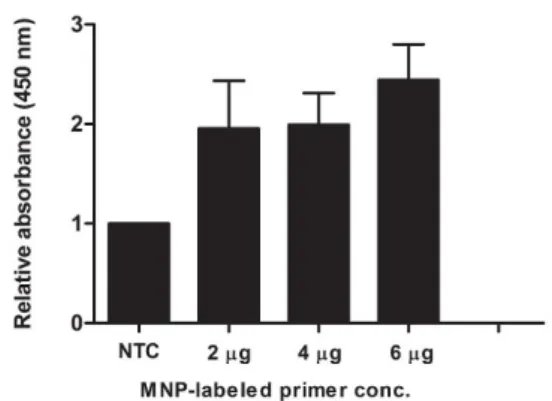
PCR for PCA3 amplification was con- ducted using 10 µM dual biotinylated reverse primer and different concentrations of MNP conjugated-forward primer. As shown in Fig- ure 2, the sample using 6 μg of MNP conju- gated-forward primer exhibited the highest OD450 ratio. At this concentration, the relative OD450 of positive sample was approximately 3-fold greater than that of negative control.
Figure 2: Optimization of primer concentration for MNP-based PCR. Each concentration (2, 4 and 6 µg) of MNP-labeled forward primer was added in the PCR reaction. After MNP-based PCR coupled with colorimetric assay, the reactions were meas- ured the OD450. The relative absorbance (450 nm) of each PCR reaction was calculated comparing to the negative control (NTC, no-target control of PCR reaction). Each bar indicates the relative ab- sorbance (mean ± SD).
Sensitivity test and detection limit
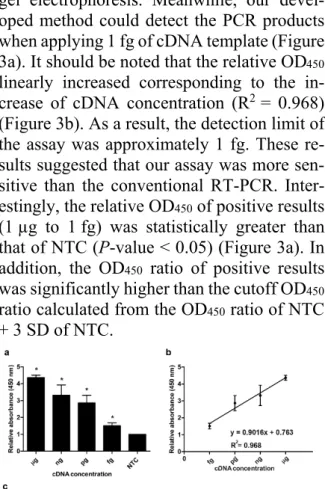
The sensitivity of the assay was investi- gated. Different concentrations of cDNA from LNCaP (1 µg to 1 fg) were employed for MNP-based PCR followed by enzyme-sub- strate detection system. The same concentra- tions of cDNA template were also used for conventional RT-PCR. As shown in Figure 3c, at lower concentration of cDNA (1 pg to 1 fg), no PCR product was visibly observed on
gel electrophoresis. Meanwhile, our devel- oped method could detect the PCR products when applying 1 fg of cDNA template (Figure 3a). It should be noted that the relative OD450
linearly increased corresponding to the in- crease of cDNA concentration (R2 = 0.968) (Figure 3b). As a result, the detection limit of the assay was approximately 1 fg. These re- sults suggested that our assay was more sen- sitive than the conventional RT-PCR. Inter- estingly, the relative OD450 of positive results (1 µg to 1 fg) was statistically greater than that of NTC (P-value < 0.05) (Figure 3a). In addition, the OD450 ratio of positive results was significantly higher than the cutoff OD450
ratio calculated from the OD450 ratio of NTC + 3 SD of NTC.
Figure 3: Sensitivity analysis (a) Relative absorb- ance (450 nm) of the MNP-based PCR coupled with colorimetric assay obtained from the different cDNA template concentrations ranging from 1 µg to 1 fg. Bars represent the relative absorbance (mean ± SD) calculated from the OD450 of each sample divided by the OD450 of control (NTC; no- target control). The t-test analysis was performed and *P-value < 0.05 compared with NTC was in- dicated. (b) Calibration curve of the relative ab- sorbance at various concentrations of cDNA tem- plate. (c) Agarose gel electrophoresis of PCR products obtained from the different cDNA tem- plate concentrations. Lane M, 100 bp ladder; Lane 1, 1 µg; Lane 2, 1 ng; Lane 3, 1pg; Lane 4, 1 fg;
NTC, no-target control.
Specificity test
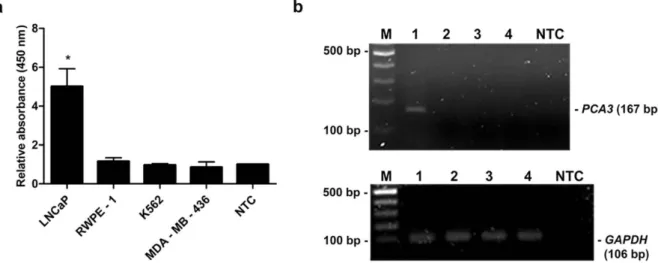
The specificity of primers for PCA3 was tested using conventional RT-PCR. The cDNA from various cell lines, i.e., RWPE-1,
LNCaP, K562 and MDA-MB-436 was indi- vidually applied as a template. Distilled water was used as a negative control (NTC). Of note, the quality of each cDNA was verified by amplification of GAPDH gene. PCR prod- ucts were analyzed by gel electrophoresis. As shown in Figure 4b, 106 bp PCR products of GAPDH were detected from the cDNA of all tested cell lines suggesting the good quality of the cDNA templates. Additionally, 167 bp PCR amplicons of PCA3 were detected only from LNCaP cell line, whereas the amplified product was not observed from RWPE-1, K562 and MDA-MB-436 cell lines (Figure 4b). Simultaneously, the MNP-based PCR was employed using MNP-labeled forward and dual biotin-labeled reverse primers. PCR products were determined by enzyme-sub- strate system. As expected, the relative OD450
of PCR products using cDNA from LNCaP cell line was significantly greater than that of PCR products from RWPE-1, K562, MDA- MB-436 cell lines and NTC (P-value < 0.05) (Figure 4a). Notably, the OD450 ratio of a pos- itive sample was higher than the OD450 ratio from the cutoff OD450 ratio of NTC + 3 SD of NTC. These results indicated that the de- signed primers were very specific to PCA3 which only expressed in LNCaP prostate can- cer cell line.
Detection of PCA3 in urine using MNP- based PCR
The capability of the assay was evaluated by detection of PCA3 in clinical specimens.
The 15 urine samples (5 biopsy-proven PCa patients, 5 BPH patients and 5 healthy sub- jects) were tested by our method and the re- sults were compared with qRT-PCR results.
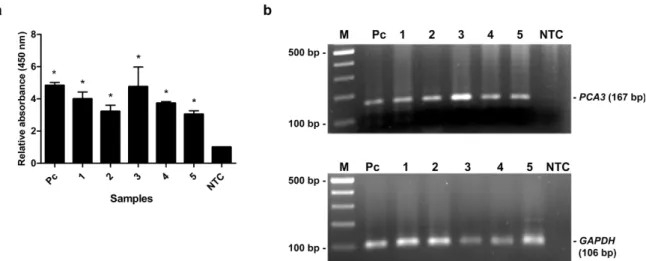
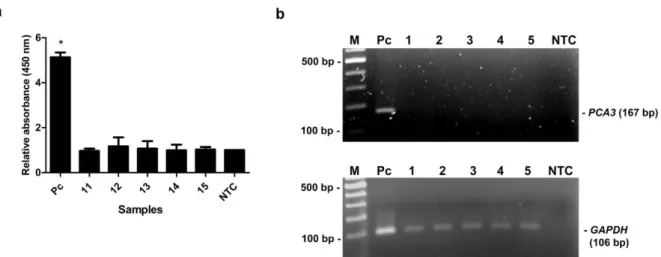
As shown in Table 2, the relative PCA3 ex- pression of PCa patients determined by qRT- PCR was remarkably higher than that of BPH patients and healthy subjects. PCR products of PCA3 and GAPDH were amplified from cDNA of individual by conventional RT-PCR to verify the PCA3 expression and DNA qual- ity. PCR products of GAPDH were amplified from all samples, while PCA3 products were detected only in PCa patients (Figure 5b). No
band was visible in other groups (Figure 6b, 7b). Using MNP-based PCR, the results demonstrated that the relative OD450 of all PCa samples was statistically higher than that of samples from BPH and healthy groups (P-
value < 0.05) (Figure 5a, 6a, 7a). Further- more, the relative OD450 of all PCa samples was greater than the cutoff OD450 ratio of NTC + 3 SD of NTC.
Figure 4: Specificity analysis. (a) Relative absorbance (450 nm) of the MNP-based PCR coupled with colorimetric assay using cDNA template obtained from various cell lines; LNCaP, RWPE-1, K562 and MDA-MB-436. Bars represent the relative absorbance (mean ± SD) calculated from the OD450 of each sample divided by the OD450 of NTC. The t-test analysis was performed and *P-value < 0.05 compared with NTC was indicated. (b) PCR products of PCA3 (upper gel) and GAPDH (lower gel) analyzed by agarose gel electrophoresis. Amplicon sizes of PCA3 and GAPDH were 167 bp and 106 bp, respec- tively. Lane M, 100 bp ladder; Lane 1-4, PCR products from LNCaP, RWPE-1, K562 and MDA-MB-436, respectively; Lane NTC, no-target control.
Table 2: Characteristics of participants Sample
no.
Age Final diagnosis Gleason score
Relative PCA3 expression level*
1 70 Prostatic adenocarcinoma 4+5 = 9 3.73 2 74 Prostatic adenocarcinoma 3+3 = 6 6.30 3 71 Prostatic adenocarcinoma 3+4 = 7 147.03 4 55 Prostatic adenocarcinoma
with bone metastasis
5+4 = 9 142.02
5 76 Prostatic adenocarcinoma 3+4 = 7 20.61
6 65 BPH - 0.40
7 79 BPH - 0.07
8 57 BPH - 0.02
9 67 BPH - 0.26
10 51 BPH (chronic inflamma- tion)
- 0.06
11 38 Normal NA -
12 34 Normal NA -
13 27 Normal NA -
14 43 Normal NA -
15 35 Normal NA -
C 63 BPH - 1.0
* Comparing to the calibrator (C)
Figure 5: The MNP-based PCR coupled with colorimetric assay of PCA3 detection from urine of PCa patients. (a) Relative absorbance (450 nm) of each PCa sample (no. 1-5). Bars represent the relative absorbance (mean ± SD) calculated from the OD450 of each sample divided by the OD450 of NTC. The t-test analysis was performed and *P-value < 0.05 compared with NTC was indicated. (b) PCR products of PCA3 (upper gel) and GAPDH (lower gel) analyzed by agarose gel electrophoresis. Amplicon sizes of PCA3 and GAPDH were 167 bp and 106 bp, respectively. Lane M, 100 bp ladder; Lane 1-5, PCR products from urine of PCa patients; Lane Pc, PCR products from LNCaP cells as a positive control;
Lane NTC, no-target control.
Figure 6: The MNP- based PCR coupled with colorimetric assay of PCA3 detection from urine of BPH patients. (a) Relative absorbance (450 nm) of each BPH sample (no. 6-10). Bars represent the relative absorbance (mean ± SD) calculated from the OD450 of each sample divided by the OD450 of NTC. The t-test analysis was performed and *P-value < 0.05 compared with NTC was indicated. (b) PCR products of PCA3 (upper gel) and GAPDH (lower gel) analyzed by agarose gel electrophoresis. Amplicon sizes of PCA3 and GAPDH were 167 bp and 106 bp, respectively. Lane M, 100 bp ladder; Lane 1- 5, PCR products from urine of BPH patients; Lane Pc, PCR products from LNCaP cells (positive control); Lane NTC, no-target control.
DISCUSSION
PCA3 is a long, non-coding prostate-spe- cific RNA, which is highly expressed in pros- tate neoplasms, but not in benign prostate dis- eases, such as BPH, prostatitis, and prostatic intraepithelial neoplasia (PIN) (Deras et al., 2008). Intriguingly, this biomarker presents in
urine. Use of the Progensa PCA3 commercial test for post-DRE urine samples was ap- proved by the US FDA in 2012 (Rittenhouse et al., 2013). Nevertheless, this assay based on target captured, transcription-mediated am- plification and hybridization is not applicable for routine screening because of its cost and complexity.
Figure 7: The MNP-based PCR coupled with colorimetric assay of PCA3 detection from urine of healthy controls. (a) Relative absorbance (450 nm) of each healthy control sample (no. 11-15). Bars represent the relative absorbance (mean ± SD) calculated from the OD450 of each sample divided by the OD450 of NTC. The t- test analysis was performed and *P- value < 0. 05 compared with NTC was indicated. (b) PCR products of PCA3 ( upper gel) and GAPDH ( lower gel) analyzed by agarose gel electrophoresis.
Amplicon sizes of PCA3 and GAPDH were 167 bp and 106 bp, respectively. Lane M, 100 bp ladder;
Lane 1- 5, PCR products from urine of healthy controls; Lane Pc, PCR products from LNCaP cells (positive control); Lane NTC, no-target control.
In this study, the MNP-based PCR in combination with colorimetric detection was successfully developed for PCA3 detection.
MNPs have advantageous properties due to their small size, high surface area, high stabil- ity, and ease of surface functionalization (Zhu et al., 2018). Functionalized MNPs coated with antibodies or oligonucleotides have been widely used for the purification, extraction and detection of biomolecules (Chan et al., 2008; Sandhu et al., 2010). Herein, the MNPs were immobilized on PCA3 forward primer.
The binding efficiency of the immobilization was approximately 51.9 %. The efficiency of immobilization is in good agreement with the result of a previous study (Tangchaikeeree et al., 2017b). In the literature, excess MNPs in- hibit the PCR reaction by adsorbing to PCR components and DNA templates and the inhi- bition was concentration-dependent (Bai et al., 2015). It has been demonstrated that the amplification of the PCR products was com- pletely inhibited when the amount of MNPs exceeded 20 μg. Therefore, the optimal con- centration of MNP conjugated-forward pri- mer was examined. As a result, positive con- trol using 6 μg of MNP conjugated-forward
primer showed the highest relative OD450 that was approximately 3-fold greater than that of negative control. There was no influence of MNPs on PCR reaction at this concentration.
Significantly, the developed method was extremely specific for the PCA3 target gene.
It has been previously evident that the expres- sion of PCA3 was found in cell lines that are androgen-dependent, such as LNCaP cells (Lemos et al., 2016). Our results showed that the relative OD450 of LNCaP was statistically higher than that of other cell lines (P-value <
0.05). Of note, there is no false positive when conducting the test using cDNA from normal prostate cell line (RWPE-1) as well as other cancer cell lines (MDA-MB-436 and K562 cell lines). As expected, the results from the MNP-based colorimetric assay are consistent with the results from gel electrophoresis.
These finding indicated that the primers for PCA3 amplification in this study are very spe- cific. In addition, the sensitivity of detection was investigated. As a result, the significant differences were observed between the rela- tive OD450 of positive samples (1 µg to 1 fg of cDNA template) and negative control. The
detection limit of the assay was approxi- mately 1 fg of the template which was 1,000- fold lower than the conventional RT-PCR fol- lowed by gel electrophoresis. These results suggested that our method is very sensitive.
The MNPs serve as a solid support to separate unbound reagents, such as free primers and reagents that are used in detection system.
Every MNP can convey thousands of copies of PCR products which in turn increase sensi- tivity. Recently, a similar technique has been applied for detection of gene expression in chronic myeloid leukemia, bacteria and para- site with the detection limits ranging from picogram to femtogram levels of template (Manthawornsiri et al., 2016; Jansaento et al., 2016; Tangchaikeeree et al., 2017b). Our re- sults are in good agreement with these previ- ous studies which indicated that this assay provides the accuracy and reliability for de- tection of the gene expression. The assay time after PCR of our method (only 1 h) was much shorter than that of previous MNP-based col- orimetric assays (>1 h) (Manthawornsiri et al., 2016; Jansaento et al., 2016;
Tangchaikeeree et al., 2017b). This could be due to our assay used the 3,3′,5,5′-tetra- methylbenzidine (TMB) as a substrate instead of 2,2'-azino-bis(3-ethylbenzothiazoline-6- sulphonic acid) (ABTS). So far as we know, ABTS is less sensitive than TMB and it may take some time to properly develop its color.
Moreover, our results showed 2-times higher relative absorbance of the colorimetric signal- ing obtained from the MNP-based PCR prod- ucts compared to previous studies (Mantha- wornsiri et al., 2016; Tangchaikeeree et al., 2017b). This significantly higher sensitivity could be granted from the advantages of high sensitive TMB substrate and using of dual bi- otin molecules, the latter of which showed that they can increase binding strength with streptavidin as reported elsewhere (Yuce et al., 2014).
To examine the potential practicality of the method, PCA3 detection in fifteen urine samples using the developed assay and the qRT-PCR were carried out. The results of our assay showed a good agreement with the
qRT-PCR results. The method could detect the significant differences of the relative OD450 of PCA3 products among PCa patients, BPH patients and healthy controls. The re- sults indicated that our assay can discriminate PCa patients from other groups. Notably, the relative PCA3 expression of PCa patients de- tected by our assay was not well correlated with the relative PCA3 expression level deter- mined by qRT-PCR. Nevertheless, our assay is feasible and applicable to use as a screening method for PCa diagnosis with high specific- ity and sensitivity. In the literature, the nano- material-based assays for PCA3 detection were recently developed. For example, the colorimetric method for PCA3 detection in urine using unmodified gold nanoparticles (AuNPs) and the thio-labeled PCR primer was established by our group (Htoo et al., 2019). In the present study, it revealed that the MNP-based colorimetric method exhibited the better sensitivity than the previous one.
Another method using AuNPs combined with the surface-enhanced Raman scattering (SERS) technique and the lateral flow assay (LFA) platform was created to detect the PCA3 mimic DNA in serum (Fu et al., 2019).
The limit of detection of the assay was esti- mated to be 3 fM. Obviously, the detection limit of our method is comparable. However, our method is simpler, cost-effective and less tedious in terms of platform preparation. In addition, it is not required for sophisticated in- strument to detect the signal.
CONCLUSION
In summary, the MNP-based PCR cou- pled with colorimetric enzyme-linked oligo- nucleotide assay has been developed. The proposed method is based on combining the MNPs-labeled forward primer and the dual biotinylated reverse primer in target gene am- plification. Subsequently, an enzyme-sub- strate system is employed for detection step.
This assay is rapid, simple and cost-effective.
The assay time after PCR can be accom- plished within an hour. Our assay provided high specificity for PCA3 when testing with PCA3 positive and negative cell lines. The
high sensitivity was also obtained with the de- tection limit at the femtogram level of tem- plate. Importantly, this assay is applicable with potential use in detection of urinary PCA3 for PCa. This technique can distinguish PCa patients from BPH patients and healthy controls based on various expression levels of PCA3 in urine sediments of subjects. It should be noted that this method demonstrated good performance compared with qRT-PCR for PCA3 detection in urine samples. Taken to- gether, the developed assay is promising and feasible for clinical practice. Finally, our as- say could be useful as a diagnostic method for screening of prostate cancer.
Conflict of interest
The authors declare that they have no fi- nancial or commercial conflict of interest.
Acknowledgments
This work was supported by National Re- search Council of Thailand and Health Sys- tems Research Institute (grant number HSRI 60-026). Khin Phyu Pyar Htoo was supported by the Mahidol-Norway Capacity Building Initiative for ASEAN Scholarship.
REFERENCES
Akbarzadeh A, Samiei M, Davaran S. Magnetic nano- particles: preparation, physical properties, and applica- tions in biomedicine. Nanoscale Res Lett. 2012;7:144.
Amiri M, Salavati-Niasari M, Akbari A. Magnetic nanocarriers: Evolution of spinel ferrites for medical applications. Adv Colloid Interface Sci. 2019;265:29- 44.
Bai Y, Cui Y, Paoli GC, Shi C, Wang D, Shi X. Nano- particles affect PCR primarily via surface interactions with PCR components: using amino-modified silica- coated magnetic nanoparticles as a main model. ACS Appl Mater Interfaces. 2015;7:13142-53.
Braconnot S, Eissa MM, Elaı̈ssari A. Morphology con- trol of magnetic latex particles prepared from oil in wa- ter ferrofluid emulsion. Colloid Polym Sci. 2013;291:
193-203.
Bussemakers MJ, van Bokhoven A, Verhaegh GW, Smit FP, Karthaus HF, Schalken JA, et al. DD3: a new prostate-specific gene, highly overexpressed in pros- tate cancer. Cancer Res. 1999;59:5975-9.
Catalona WJ. Prostate cancer screening. Med Clin North Am. 2018;102:199-214.
Catalona WJ, Richie JP, Ahmann FR, Hudson MA, Scardino PT, Flanigan RC, et al. Comparison of digital rectal examination and serum prostate specific antigen in the early detection of prostate cancer: results of a multicenter clinical trial of 6,630 men. J Urol. 1994;
151:1283-90.
Chan CP, Cheung Y, Renneberg R, Seydack M. New trends in immunoassays. Adv Biochem Eng Biotech- nol. 2008;109:123-54.
Cucchiara V, Cooperberg MR, Dall'Era M, Lin DW, Montorsi F, Schalken JA, et al. Genomic markers in prostate cancer decision making. Eur Urol. 2018;73:
572-82.
Cui Y, Cao W, Li Q, Shen H, Liu C, Deng J, et al. Eval- uation of prostate cancer antigen 3 for detecting pros- tate cancer: a systematic review and meta-analysis. Sci Rep. 2016;6:25776.
Deras IL, Aubin SMJ, Blase A, Day JR, Koo S, Partin AW, et al. PCA3: a molecular urine assay for predict- ing prostate biopsy outcome. J Urol. 2008;179:1587- 92.
Fu X, Wen J, Li J, Lin H, Liu Y, Zhuang X, et al.
Highly sensitive detection of prostate cancer specific PCA3 mimic DNA using SERS-based competitive lat- eral flow assay. Nanoscale. 2019;11:15530-6.
Htoo KPP, Yamkamon V, Yainoy S, Suksrichavalit T, Viseshsindh W, Eiamphungporn W. Colorimetric de- tection of PCA3 in urine for prostate cancer diagnosis using thiol-labeled PCR primer and unmodified gold nanoparticles. Clin Chim Acta. 2019;488:40-9.
Jangpatarapongsa K, Polpanich D, Yamkamon V, Dit- tharot Y, Peng-On J, Thiramanas R, et al. DNA detec- tion of chronic myelogenous leukemia by magnetic na- noparticles. Analyst. 2011;136:354-8.
Jansaento W, Jangpatarapongsa K, Polpanich D, Won- glumsom W. Detection of Campylobacter DNA using magnetic nanoparticles coupled with PCR and a color- imetric end-point system. Food Sci Biotechnol. 2016;
25:193-8.
Kim C, Searson PC. Detection of Plasmodium lactate dehydrogenase antigen in buffer using aptamer-modi- fied magnetic microparticles for capture, oligonucleo- tide-modified quantum dots for detection, and oligonu- cleotide-modified gold nanoparticles for signal ampli- fication. Bioconjug Chem. 2017;28:2230-4.
Lemos AEG, Ferreira LB, Batoreu NM, de Freitas PP, Bonamino MH, Gimba ER. PCA3 long noncoding RNA modulates the expression of key cancer-related genes in LNCaP prostate cancer cells. Tumour Biol.
2016;37:11339-48.
Loeb S, Partin AW. Review of the literature: PCA3 for prostate cancer risk assessment and prognostication.
Rev Urol. 2011;13:e191-5.
Ma X, Huang J. Predicting clinical outcome of therapy- resistant prostate cancer. Proc Natl Acad Sci USA.
2017;116:11090-2.
Manthawornsiri Y, Polpanich D, Yamkamon V, Thira- manas R, Hongeng S, Rerkamnuaychoke B, et al. Mag- netic nanoparticles PCR enzyme-linked gene assay for quantitative detection of BCR/ABL fusion gene in chronic myelogenous leukemia. J Clin Lab Anal. 2016;
30:534-42.
Mody VV, Cox A, Shah S, Singh A, Bevins W, Parihar H. Magnetic nanoparticle drug delivery systems for targeting tumor. Appl Nanosci. 2014;4:385-92.
Montagne F, Mondain-Monval O, Pichot C, Mozza- nega H, Elaı̈ssari A. Preparation and characterization of narrow sized (o/w) magnetic emulsion. J Magn Magn Mater. 2002;250:302-12.
Nakajima N, Ikada Y. Mechanism of amide formation by carbodiimide for bioconjugation in aqueous media.
Bioconjug Chem. 1995;6:123-30.
Perez JW, Vargis EA, Russ PK, Haselton FR, Wright DW. Detection of respiratory syncytial virus using na- noparticle amplified immuno-polymerase chain reac- tion. Anal Biochem. 2011;410:141-8.
Promkan M, Dakeng S, Chakrabarty S, Bögler O, Pat- masiriwat P. The effectiveness of cucurbitacin B in BRCA1 defective breast cancer cells. PLoS One. 2013;
8:e55732.
Rittenhouse H, Blase A, Shamel B, Schalken J, Gros- kopf J. The long and winding road to FDA approval of a novel prostate cancer test: our story. Clin Chem.
2013;59:32-4.
Roobol MJ, Zappa M, Määttänen L, Ciatto S. The value of different screening tests in predicting prostate biopsy outcome in screening for prostate cancer data from a multicenter study (ERSPC). Prostate. 2007;67:
439-46.
Sandhu A, Handa H, Abe M. Synthesis and applica- tions of magnetic nanoparticles for biorecognition and point of care medical diagnostics. Nanotechnology.
2010;21:442001.
Shabestari Khiabani S, Farshbaf M, Akbarzadeh A, Davaran S. Magnetic nanoparticles: preparation meth- ods, applications in cancer diagnosis and cancer ther- apy. Artif Cells Nanomed Biotechnol. 2017;45:6-17.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018.
CA Cancer J Clin. 2018;68:7-30.
Smith RA, Cokkinides V, Eyre HJ, American Cancer Society. American Cancer Society guidelines for the early detection of cancer, 2004. CA Cancer J Clin.
2004;54:41-52.
Suangtamai T, Tanyong DI. Diallyl disulfide induces apoptosis and autophagy via mTOR pathway in mye- loid leukemic cell line. Tumour Biol. 2016;37:10993- 9.
Taitt HE. Global trends and prostate cancer: A review of incidence, detection, and mortality as influenced by race, ethnicity, and geographic location. Am J Mens Health. 2018;12:1807-23.
Tangchaikeeree T, Polpanich D, Bentaher A, Baraket A, Errachid A, Agusti G, et al. Combination of PCR and dual nanoparticles for detection of Plasmodium falciparum. Colloids Surf B, Biointerfaces. 2017a;159:
888-97.
Tangchaikeeree T, Sawaisorn P, Somsri S, Polpanich D, Putaporntip C, Tangboriboonrat P, et al. Enhanced sensitivity for detection of Plasmodium falciparum ga- metocytes by magnetic nanoparticles combined with enzyme substrate system. Talanta. 2017b;164:645-50.
Thompson IM, Pauler DK, Goodman PJ, Tangen CM, Lucia MS, Parnes HL, et al. Prevalence of prostate can- cer among men with a prostate-specific antigen level <
or =4.0 ng per milliliter. N Engl J Med. 2004;350:
2239-46.
US Preventive Services Task Force, Grossman DC, Curry SJ, Owens DK, Bibbins-Domingo K, Caughey AB, et al. Screening for prostate cancer: US preventive services task force recommendation statement. JAMA.
2018;319:1901-13.
Wang Y, Liu XJ, Yao XD. Function of PCA3 in pros- tate tissue and clinical research progress on developing a PCA3 score. Chin J Cancer Res. 2014;26:493-500.
Yuce M, Kurt H, Budak H. Characterization of a dual biotin tag for improved single stranded DNA produc- tion. Anal Methods. 2014;6:548-7.
Zhu N, Ji H, Yu P, Niu J, Farooq MU, Akram MW, et al. Surface modification of magnetic iron oxide nano- particles. Nanomaterials (Basel). 2018;8(10):810.