Zeitschrift für Kristallographie New Crystal Structures 212, 2 4 5 - 2 4 6
© by R. Oldenbourg Verlag, München 1997
245
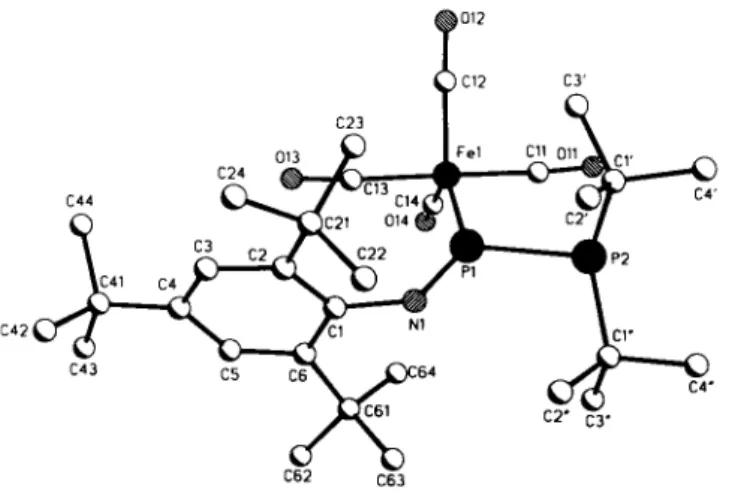
Crystal structure of tetracarbonyl^ 1 -l,l-di-terí-butyl-3-(2,4,6-tri-terí- butylphenyl)-l,2-diphosphatriaz-2-ene)iron(0), C3oH47FeN04P2
M. Nieger, E. Niecke and M. Lysek
Universität Bonn, Institut für Anorganische Chemie, Gerhard-Domagk-Str. I, D-53121 Bonn, Germany Received September 2, 1996, CSD-No. 402616
Table 2. Final atomic coordinates and displacement parameters (in Â2)
«2 C63
Source of material: source of material: see ref. 1.
The diphosphatriazene is η'-coordinated by the lone pair of the trigonal planar configurated imino-phosphorus to a tetracarbonyl iron fragment. The bulky iminophosphane ligand adopts an equa- torial coordination site of the trigonal bipyramidal coordinated iron. The P-Fe bond (216.5(1) pm) is significant shorter than the P-Fe bond (221.5(1) pm) in a comparable diphosphene iron complex (see ref. 2). The P=N double bond (156.5(2) pm) is similar to that found in the uncomplexed compound (157.8(2) pm (see ref. 2)). The phosphino and the aryl ligand adopt a trans configuration relative to the P=N double bond (torsion angle C( 1 )-N( 1 )-P( 1 )-P(2) 179.0(2)°). For the chemistry of iminophos- phanes see ref. 3.
C30H47FeNO4P2, triclinic, PI (No. 2), a =10.720(1) Â,
b =11.371(1) Â, c =14380(1) Â, α =80.63(1)°, β =7631(1)°, γ=81.00(1)°, V=1667.6 Â3
, Ζ =2,
R(F)=0.045,
Rvi(r,all data) =0.123.
Table 1. Parameters used for the X-ray data collection
Crystal: red block, size 0.40 χ 0.40 χ 0.55 mm Wavelength: Mo Ka radiation (0.71069 À)
μ: 5.80 cm"1
Diffractometer: Stoe-Siemens AED
Scan mode: profile fitting mode (see ref. 4)
Τ measurement" 293 Κ
28max: 50°
Ν (hkl)wúque' 5840
Criterion for /0: Io > 2 σ(/0)
Ν (pararnirtfinnT· 338
Programs: SHELXS-86, SHELXL-93
Atom Site Occ. X y ζ t/iso
H(3) 2i 0.3940(3) 0.6351(2) 0.1764(2) 0.054 H(5) 2i 0.2385(3) 0.4422(2) 0.4184(2) 0.062 H(22A) 2 i 0.7208(3) 0.6067(3) 0.2402(2) 0.087 H(22B) 2i 0.8193(3) 0.5881(3) 0.1426(2) 0.087 H(22C) 2 i 0.7814(3) 0.4768(3) 0.2184(2) 0.087 H(23A) 2 i 0.7034(3) 0.3843(3) 0.0915(2) 0.086 H(23B) 2 i 0.7537(3) 0.4930(3) 0.0176(2) 0.086 H(23C) 2 i 0.6105(3) 0.4671(3) 0.0321(2) 0.086 H(24A) 2i 0.5757(3) 0.7283(3) 0.1399(2) 0.095 H(24B) 2i 0.5344(3) 0.6723(3) 0.0608(2) 0.095 H(24C) 21 0.6776(3) 0.6981(3) 0.0463(2) 0.095 C(42) 2i 0.820(3) 0.1738(5) 0.7543(4) 0.2915(4) 0.084(1) H(42A) 2i 0.820(3) 0.0918(5) 0.8013(4) 0.2888(4) 0.126 H(42B) 2i 0.820(3) 0.2380(5) 0.7817(4) 0.2367(4) 0.126 H(42C) 2i 0.820(3) 0.1983(5) 0.7630(4) 0.3498(4) 0.126 C(43) 2i 0.820(3) 0.0533(4) 0.5865(4) 0.3738(3) 0.078(1) H(43A) 2 i 0.820(3) 0.0447(4) 0.5036(4) 0.3744(3) 0.117 H(43B) 2 i 0.820(3) -0.0261(4) 0.6352(4) 0.3661(3) 0.117 H(43C) 2 í 0.820(3) 0.0726(4) 0.5970(4) 0.4337(3) 0.117 C(44) 2 i 0.820(3) 0.1232(5) 0.6107(4) 0.1973(3) 0.083(1) H(44A) 2i 0.820(3) 0.1164(5) 0.5277(4) 0.1963(3) 0.125 H(44B) 2 i 0.820(3) 0.1872(5) 0.6382(4) 0.1424(3) 0.125 H(44C) 2 i 0.820(3) 0.0411(5) 0.6576(4) 0.1950(3) 0.125 C(42') 2 i 0.180(3) 0.054(2) 0.553(2) 0.301(2) 0.084(1) H(42D) 2 i 0.180(3) -0.024(2) 0.606(2) 0.298(2) 0.126 H(42E) 2i 0.180(3) 0.043(2) 0.503(2) 0.363(2) 0.126 H(42F) 2i 0.180(3) 0.073(2) 0.503(2) 0.251(2) 0.126 C(43·) 2 i 0.180(3) 0.167(2) 0.723(1) 0.203(1) 0.078(1) H(43D) 2i 0.180(3) 0.239(2) 0.767(1) 0.198(1) 0.117 H(43E) 2i 0.180(3) 0.088(2) 0.776(1) 0.212(1) 0.117 H(43F) 2i 0.180(3) 0.178(2) 0.687(1) 0.146(1) 0.117 C(44') 2i 0.180(3) 0.129(2) 0.698(2) 0.379(1) 0.083(1) H(44D) 2/ 0.180(3) 0.197(2) 0.746(2) 0.375(1) 0.125 H(44E) 2i 0.180(3) 0.120(2) 0.643(2) 0.438(1) 0.125 H(44F) 2i 0.180(3) 0.049(2) 0.749(2) 0.378(1) 0.125 H(62A) 2i 0.2383(5) 0.3018(4) 0.5243(2) 0.155 H(62B) 2 i 0.3016(5) 0.3961(4) 0.5606(2) 0.155 H(62C) 2i 0.3214(5) 0.2600(4) 0.6027(2) 0.155 H(63A) 2 i 0.5398(4) 0.4008(3) 0.5131(2) 0.127 H(63B) 2i 0.6280(4) 0.2992(3) 0.4581(2) 0.127 H(63C) 2 i 0.5508(4) 0.2680(3) 0.5642(2) 0.127 H(64A) 2 i 0.3736(4) 0.1519(3) 0.4258(3) 0.120 H(64B) 2 i 0.4476(4) 0.1133(3) 0.5100(3) 0.120 H(64C) 2i 0.5248(4) 0.1445(3) 0.4038(3) 0.120 H(2'l) 2 i 1.0654(4) 0.3141(3) 0.1654(3) 0.119 H(2'2) 2i 0.9463(4) 0.3613(3) 0.1182(3) 0.119 H(2'3) 2 i 1.0826(4) 0.3268(3) 0.0531(3) 0.119 H(3'l) li 0.9117(3) 0.0870(3) 0.0461(2) 0.104 H(3'2) 21 0.9881(3) 0.1874(3) -0.0205(2) 0.104 H(3'3) 21 0.8518(3) 0.2220(3) 0.0446(2) 0.104 H(4'l) 21 1.1027(3) 0.0119(4) 0.1155(3) 0.127 H(4'2) 2/ 1.1669(3) 0.0984(4) 0.1594(3) 0.127
246
C30H47FeNO4P2Table 2. (Continued) Table 2. (Continued)
Atom Site Occ. χ Atom Site Occ. χ Uis
H(4'3) li 1.1760(3) H(2" 1 ) 21 0.9726(4) H(2"2) 21 0.9400(4) H(2"3) 2/ 0.8287(4) H(3"l) 2/ 0.8537(5)
0.1136(4) 0.0478(3) 0.127 0.3354(3) 0.3047(3) 0.122 0.3036(3) 0.4173(3) 0.122 0.3285(3) 0.3603(3) 0.122 0.0121(3) 0.4262(3) 0.132
H(3"2) 21 0.7553(5) H(3"3) 2/ 0.8667(5) H(4" 1 ) 2 i 1.0830(4) H(4"2) 2i 1.0929(4) H(4"3) 2/ 1.1300(4)
0.1293(3) 0.4354(3) 0.132 0.1044(3) 0.4924(3) 0.132 0.0239(4) 0.3415(3) 0.149 0.1169(4) 0.4081(3) 0.149 0.1484(4) 0.2955(3) 0.149
Table 3. Final atomic coordinates and displacement parameters (in À2)
Atom Site χ y ζ Ut ι Un t/33 Un U13 I/23 Fe(l) li 0.57717(4) 0.10335(3) 0.17566(3) 0.0465(2) 0.0438(2) 0.0576(3) -0.0073(2) -0.0133(2) -0.0109(2) P(l) 2i 0.68683(6) 0.20528(5) 0.23527(5) 0.0424(4) 0.0343(3) 0.0392(3) -0.0045(3) -0.0077(3) -0.0047(3) P(2) 2/ 0.88747(7) 0.11906(6) 0.24007(5) 0.0492(4) 0.0378(4) 0.0581(4) -0.0023(3) -0.0184<3) -0.0052(3) N(I) 21 0.6501(2) 0.3233(2) 0.2837(1) 0.055(1) 0.036(1) 0.037(1) -0.0041(9) -0.0076(9) -0.0032(8) C(l) 21 0.5315(3) 0.4010(2) 0.2919(2) 0.055(2) 0.034(1) 0.035(1) -0.004(1) -0.005(1) -0.007(1) C(2) li 0.5219(2) 0.5035(2) 0.2212(2) 0.048(1) 0.038(1) 0.037(1) -0.007(1) -0.004(1) -0.006(1) C(3) 2 Í 0.4015(3) 0.5705(2) 0.2244(2) 0.049(2) 0.038(1) 0.043(1) -0.003(1) -0.005(1) -0.003(1) C(4) li 0.2921(3) 0.5475(2) 0.2942(2) 0.049(2) 0.043(1) 0.044(1) -0.009(1) 0.000(1) -0.013(1) C(5) 2/ 0.3093(3) 0.4561(2) 0.3681(2) 0.059(2) 0.046(2) 0.042(2) -0.009(1) 0.008(1) -0.010(1) C(6) li 0.4254(3) 0.3837(2) 0.3718(2) 0.065(2) 0.038(1) 0.037(1) -0.007(1) 0.002(1) -0.005(1) C(21) 2/ 0.6380(3) 0.5464(2) 0.1436(2) 0.046(1) 0.039(1) 0.041(1) -0.004(1) -0.003(1) 0.002(1) C(22) 21 0.7505(3) 0.5553(3) 0.1906(2) 0.054(2) 0.056(2) 0.061(2) -0.014(1) -0.008(1) -0.002(1) C(23) 21 0.6804(3) 0.4651(3) 0.0637(2) 0.064(2) 0.059(2) 0.039(1) -0.004(1) 0.002(1) -0.003(1) C(24) 21 0.6031(3) 0.6732(3) 0.0929(2) 0.056(2) 0.052(2) 0.066(2) -0.005(1) 0.004(2) 0.014(1) C(41) li 0.1631(3) 0.6242(3) 0.2900(2) 0.047(2) 0.056(2) 0.056(2) -0.008(1) -0.001(1) -0.014(1) C(61) 2 i 0.4348(3) 0.2955(3) 0.4656(2) 0.090(2) 0.046(2) 0.038(2) -0.003(2) 0.007(2) 0.003(1) C(62) li 0.3125(5) 0.3152(4) 0.5459(2) 0.135(4) 0.084(3) 0.048(2) 0.020(2) 0.028(2) 0.012(2) C(63) 2 i 0.5489(4) 0.3179(3) 0.5038(2) 0.131(4) 0.070(2) 0.048(2) 0.003(2) -0.024(2) 0.000(2) C(64) li 0.4463(4) 0.1641(3) 0.4498(3) 0.110(3) 0.047(2) 0.064(2) -0.013(2) 0.011(2) 0.007(2) C(l') li 0.9950(3) 0.1797(3) 0.1225(2) 0.042(2) 0.056(2) 0.064(2) 0.001(1) -0.006(1) -0.011(1) C(2·) li 1.0251(4) 0.3071(3) 0.1140(3) 0.071(2) 0.064(2) 0.094(3) -0.019(2) 0.003(2) -0.005(2) C(3·) li 0.9306(3) 0.1679(3) 0.0405(2) 0.058(2) 0.088(2) 0.055(2) 0.004(2) -0.004(2) -0.015(2) C(4') li 1.1222(3) 0.0926(4) 0.1101(3) 0.053(2) 0.091(3) 0.101(3) 0.017(2) -0.011(2) -0.022(2) C(l") li 0.9321(3) 0.1619(3) 0.3478(2) 0.080(2) 0.062(2) 0.063(2) -0.005(2) -0.040(2) 0.000(2) C(2") li 0.9169(4) 0.2946(3) 0.3585(3) 0.117(3) 0.073(2) 0.074(2) -0.020(2) -0.053(2) -0.012(2) C(3") li 0.8437(5) 0.0958(3) 0.4335(3) 0.129(4) 0.076(2) 0.060(2) -0.008(2) -0.035(2) 0.007(2) C(4") li 1.0727(4) 0.1077(4) 0.3483(3) 0.096(3) 0.109(3) 0.106(3) 0.000(2) -0.066(3) 0.001(3) C ( l l ) li 0.6962(3) -0.0272(3) 0.1825(3) 0.069(2) 0.044(2) 0.086(2) -0.011(2) -0.026(2) -0.017(2) 0(11) li 0.7622(3) -0.1156(2) 0.1839(2) 0.098(2) 0.047(1) 0.163(3) 0.005(1) -0.053(2) -0.032(2) C(12) li 0.6006(3) 0.1029(3) 0.0479(3) 0.070(2) 0.066(2) 0.072(2) 0.012(2) -0.023(2) -0.022(2) 0(12) li 0.6192(3) 0.1014(3) -0.0333(2) 0.124(2) 0.129(3) 0.067(2) 0.043(2) -0.031(2) -0.036(2) C(13) li 0.4490(3) 0.2281(3) 0.1732(2) 0.052(2) 0.060(2) 0.065(2) -0.010(2) -0.017(1) -0.016(1) 0(13) li 0.3629(2) 0.2986(2) 0.1680(2) 0.063(1) 0.079(2) 0.104(2) 0.014(1) -0.034(1) -0.032(1) C(14) li 0.4586(4) 0.0104(3) 0.2474(3) 0.064(2) 0.064(2) 0.106(3) -0.020(2) -0.019(2) -0.004(2) 0(14) li 0.3854(3) -0.0481(3) 0.2922(3) 0.090(2) 0.103(2) 0.179(3) -0.053(2) -0.008(2) 0.022(2)
References
1. Lysek, M.: Aminodiphosphene und Iminophosphane mit sterisch an- spruchsvollen Stickstoffsubstituenten. Dissertation, Universität Bielefeld, Germany 1987.
2. Cowley, A. H.; Kilduff, J. E.; Lasch, J. G.; Norman, N. C ; Pakulski, M.;
Ando, F.; Wright, T. C.: Reactivity of Diphosphenes and Phosphaarsenes toward Metal Carbonyls. J. Am. Chem. Soc. 105 (1983) 7751-7752.
3. Niecke, E.; Gudat, D.: Iminophosphanes: Unconventional Compounds of Main Group Elements. Angew. Chem. Int. Ed. Engl. 30 (1991) 217-237.
4. Clegg, W.: Faster Data Collection Without Loss of Precession. An Exten- sion of the Leamt Profile Method. Acta Crystallogr. A37 (1981) 22-28.
4. Sheldrick, G. M.: SHELXS-86. Program for the solution of crystal struc- tures. University of Göttingen, Germany 1986.
5. Sheldrick, G. M.: SHELXL-93. Program for refining crystal structures.
University of Göttingen, Germany 1993.