Z. Kristallogr. NCS 219 (2004) 371-372
© by Oldenbourg Wissenschaftsverlag, München
371
Crystal structure of (?7 7 -2,4,6-cycloheptatrien-l-ylium)(?/ 5 -l,2,3,4,5-penta- methy 1-2,4-cyclopentadien-1 -yl)chromium(I) hexafluorophosphate,
[Cr(C 7 H7)(CioH 15 )][PF 6 ]
A. Hradsky, H. Kopacka, B. Bildstein and K. Wurst*
University of Innsbruck, Institute of General, Inorganic and Theoretical Chemistry, Innrain 52a, 6020 Innsbruck, Austria Received April 13, 2004, accepted and available on-line September 13, 2004; CCDC no. 1267/1305
C<7)
C ( 7 A Ï CI6)
C I 0 3 A
F ( 2 )
Abstract
CnH
22CrF6P, orthorhombic,
Amal(no. 40),
a= 15.9186(9) A,
b = 15.6388(4) A , c = 7.2709(3) A , V= 1810.1 A3
, Z = 4,
Rei(F)
= 0.035,
wRnftF2)= 0.084,
T=213 K.
Source of material
The Cr complex was obtained from a reaction of lithium penta- methylcyclopentadienide, chromium(IH)chloride, cyclohepta- triene and magnesium powder in tetrahydrofuran solution, followed by aqueous workup with sodium hexafluorophosphate solution [1].
Experimental details
The compound crystallizes as a racemic twin in a ratio of 0.54(4):
0.44 determined by the Flack x parameter refined as twinning part. The hydrogen atoms of the planar cycloheptatrienylium ring were found and refined with bond restraints
(dc—h= 0.97 A).
Discussion
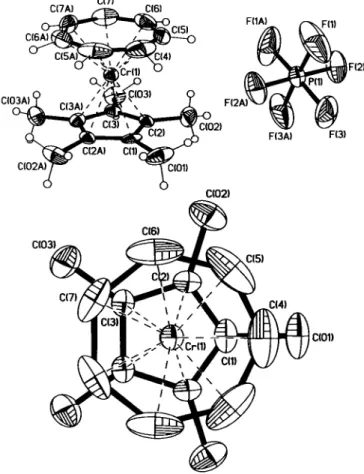
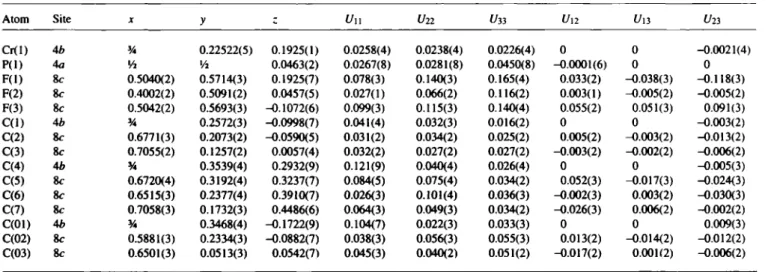
The asymmetric unit contains half a molecule of the salt. The Crl, C1 and C4 atoms of the cation lie in a mirror plane, whereas the P1 atom of the anion lies on the two-fold axis. The Cr—C distances to the 5-membered ring axe between 2.183A-2.184A with a dis- tance of Crl to the plane of 1.814 A . For the 7-membered ring the Cr—C distances lie between 2.140 A and 2.151 A , whereas the distance of Crl to the plane is much shorter with a value of 1.424 A . The averaged C—C distances of the 5- and 7-membered rings are 1.428 A and 1.393 A , respectively. The structure shows two interesting details of the coordination to the 7-membered ring. First, as shown in the figure below, the mobility is around two times higher compared with the other ring. Second, the C-H bonds are not in the plane of the ring, they are bent by approxi- mately 10° in direction to the chromium ion.
Positional disorder of the 7-ring can be excluded due to the ab- sence of rest electron density appearing between the carbon atoms, if only a major part of disordered model was considered.
Refinements on data collected at 213 K, 243 K and 293 K gave solely hints for continuously increasing libration.
Table 1. Data collection and handling.
Crystal:
Wavelength:
P-
Diffractometer, scan mode:
20max'-
WlJWVeasurcd, W^iunique:
C r i t e r i o n f o r lobs, N(hkl)p.
Nfparamhtfined:
Programs:
yellow prism, size 0.07 x 0.08 x 0.11 mm Mo Ka radiation (0.71073 Â)
7.75 cm"'
Nonius KappaCCD, <p/a>
48°
4678, 1477
/obs > 2 a(Idx), 1 3 8 0
146
SHELXS-97 [2], SHELXL-97 [3], SHELXTL [4]
Table 2. Atomic coordinates and displacement parameters (in Â2).
* Correspondence author (e-mail: klaus.wurst@uibk.ac.at)
Atom Site X y z l/i*>
H(4) 4b % 0.404(5) 0.21(1) 0.15(4)
H(5) 8c 0.621(2) 0.347(3) 0.281(7) 0.08(2) H(6) 8c 0.591(1) 0.230(4) 0.386(9) 0.09(2) H(7) 8c 0.681(4) 0.118(2) 0.466(8) 0.09(2)
H(01A) 4b % 0.343(3) -0.304(3) 0.05(2)
H(01B) 8c 0.699(2) 0.376(3) -0.133(7) 0.07(2)
H(02A) 8c 0.5687 0.2117 -0.2058 0.075
H(02B) 8c 0.5842 0.2953 -0.0875 0.075
H(02C) 8c 0.5535 0.2101 0.0095 0.075
H(03A) 8c 0.6784 0.0154 0.1436 0.068
H(03B) 8c 0.6383 0.0183 -0.0557 0.068
H(03C) 8c 0.5979 0.0722 0.1059 0.068
372
[Cr(C7H7)(CioHi5)][PF6]Table 3. Atomic coordinates and displacement parameters (in À2).
Atom Site X y : Uu 1/22 i/33 Un Un U21
Cr(l) 4b % 0.22522(5) 0.1925(1) 0.0258(4) 0.0238(4) 0.0226(4) 0 0 -0.0021(4)
P(l) \a Yi Vi 0.0463(2) 0.0267(8) 0.0281(8) 0.0450(8) -0.0001(6) 0 0
F(l) 8c 0.5040(2) 0.5714(3) 0.1925(7) 0.078(3) 0.140(3) 0.165(4) 0.033(2) -0.038(3) -0.118(3) F(2) 8c 0.4002(2) 0.5091(2) 0.0457(5) 0.027(1) 0.066(2) 0.116(2) 0.003(1) -0.005(2) -0.005(2) F(3) 8c 0.5042(2) 0.5693(3) -0.1072(6) 0.099(3) 0.115(3) 0.140(4) 0.055(2) 0.051(3) 0.091(3)
C(l) 4b % 0.2572(3) -0.0998(7) 0.041(4) 0.032(3) 0.016(2) 0 0 -0.003(2)
C(2) 8c 0.6771(3) 0.2073(2) -0.0590(5) 0.031(2) 0.034(2) 0.025(2) 0.005(2) -0.003(2) -0.013(2) C(3) 8c 0.7055(2) 0.1257(2) 0.0057(4) 0.032(2) 0.027(2) 0.027(2) -0.003(2) -0.002(2) -0.006(2)
C(4) 4b V* 0.3539(4) 0.2932(9) 0.121(9) 0.040(4) 0.026(4) 0 0 -0.005(3)
C(5) 8c 0.6720(4) 0.3192(4) 0.3237(7) 0.084(5) 0.075(4) 0.034(2) 0.052(3) -0.017(3) -0.024(3) C(6) 8c 0.6515(3) 0.2377(4) 0.3910(7) 0.026(3) 0.101(4) 0.036(3) -0.002(3) 0.003(2) -0.030(3) C(7) 8c 0.7058(3) 0.1732(3) 0.4486(6) 0.064(3) 0.049(3) 0.034(2) -0.026(3) 0.006(2) -0.002(2)
C(01) 4b % 0.3468(4) -0.1722(9) 0.104(7) 0.022(3) 0.033(3) 0 0 0.009(3)
C(02) 8c 0.5881(3) 0.2334(3) -0.0882(7) 0.038(3) 0.056(3) 0.055(3) 0.013(2) -0.014(2) -0.012(2) C(03) 8c 0.6501(3) 0.0513(3) 0.0542(7) 0.045(3) 0.040(2) 0.051(2) -0.017(2) 0.001(2) -0.006(2)
References
1. Hradsky, A.: Pentamethylferrocene. PhD Dissertation, University of Innsbruck, Austria 1996.
2. Sheldrick, G. M.: SHELXS-97. Program for the Solution of Crystal Structure. University of Gottingen, Germany 1997.
3. Sheldrick, G. M.: SHELXL-97. Program for the Refinement of Crystal Structure. University of Gottingen, Germany 1997.
4. Sheldrick, G. M.: SHELXTL. Version 5.1. Programs for the Analysis of Crystal Structures. Bruker Analytical X-Ray Instruments Inc., Madison WI53719, USA 1997.