ζ . Kristallogr. NCS 213 (1998) 4 7 3
© by R. Oldenbourg Verlag, München
Crystal structure of a-tripotassium bismuthide, а-КзВ!, a revision
H. K e r b e r , H . - J . D e i s e r o t h a n d R . W a l t h e r
Universitäl-GH Siegen, Anorganische Chemie. D-57068 Siegen, Germany
Received Febniary 3, 1998, transferred to 2nd update of database ICSD in 1998, CSD-No. 409223
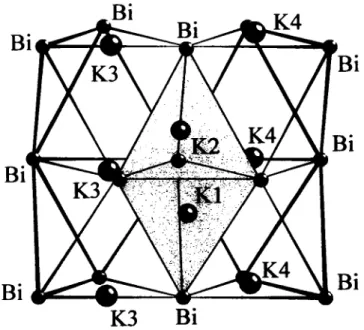
In contrast to ß-KsBi (see réf. 1 ), the crystal structure of the low temperature phase а - К з В ! (Tu = 5 5 0 К ) is isotypic to the modi- fied N a s A s type ( see refs. 2 , 3 ) , recently determinded on the basis of a larger unit cell than originally proposed for N a s A s (see ref.
4). The arrangement of the A s atoms coiresponds to the topology of a hexagonal close packing with K1 and K2 in neighbored, face sharing tetrahedral holes ( d ( K l a - K l b ) = 371.6 pm). КЗ and K4 are approximately trigonally coordinated by Bi with different diplacements fnjm the trigonal plane (КЗ: 2 2 pm, K4: 34 pm). A slight anisotropy of the К atoms is noticed (see ref. 5).
BÌK3. hexagonal, Рвзст (No. 185), a =10.649(2) Â,
с = 1 0 . 9 4 0 ( 2 ) Â ,
ν=10Ί4Λ
 ^ Z = 6 , pm = 3 . 0 2 6 g cm"^R(F) =0.032, R^F^) = 0 . 0 7 5 .
Table 1. Parameters used for the X-ray data collection
КЗ Bi
Source of material: Single crystals as well as pure microcrystal- line samples of purple-black, air sensitive K3BÌ with metallic lustre can be prepared from a stoichiometric mixture of the elements in the presence o f a slight e x c e s s of potassium. The reaction has to be carried out in a sealed V 2 A steel ampoule by repeated annealing at Τ = 7 7 3 К and final evaporation of the excess potassium under vacuum conditions.
Table 2. Final atomic coordinates and displacement parameters (in Â^)
Ciystal: ршр1е-Ь1аск plate, size 0.1 χ 0.1 χ .0.5 mm Wavelength: Mo Ka radiation (0.71069 Â)
μ: 262.19 cm-'
Diffractometen Enraf-Nonius CAD4
Scan mode: e m
'Îmeasuremenl'· 293 К
60.04°
ЩНкОипцие: 1087
Criterion for /0: Io >2 ado)
Ν(ραΓαιη)Γφκά·· 26
Program: SHELXL-93
Atom Site X ζ í/ll t/22 ί/33 Un Un Uli
Bi 6c 0.3321(2) X 0.7499(9) 0.0386(4) í/ll 0.0344(2) 0.0216(6) -0.0015(3) Un
K(l) 6c 0.313(1) X 0.0841(5) 0.099(7) и и 0.041(4) 0.069(7) -0.001(4) Un
K(2) 6c 0.3547(7) X 0.4193(5) 0.065(3) Un 0.046(4) 0.022(2) 0.011(3) Un
K(3) 4b 1/3 2/3 0.230(2) 0.045(2) Un 0.15(1) 0.023(1) 0 0
K(4) 2a 0 0 0.281(2) 0.041(4) Un 0.14(2) 0.021(2) 0 0
References
1. Sands,D.E.; Wood,D.H.;Ramsey,W.J.:Theciystalstructureofß-K3Bi.
Acta Crystallogr. 16 (1963) 316-319.
2. Hafner, P.; Range, K.-J.: NasAs revisited: high pressure synthesis of single crystals and structure refinement. J. Alloys Compd. 216 (1996) 7-10.
3. Range, K.-J.; Ehrl, R.; Hafner, P.: NasAs revisited II. Space group confirmation and structure refinement at 150, 210 and 240 K. J. Alloys Compd. 240(1996) 19-24.
4. Brauer, G.; Zintl, В.: Konstitution von Phosphiden, Arseniden, Anti- moniden und Wismutiden des Lithiums, Natriums und Kaliums. Z. Phys.
Chem. 37 (1937) 323-352.
Kerber, H.: Beiträge zur Festkörpeichemie von Trialkalimetallpniktiden und Clusterverbindungen mit Indium. Dissertation, Universität Gesamt- 3hochschuIe Siegen, Germany 1997.
Sheldrick, G. M.: SHELXL-93. Program for refining crystal structures.
University of Göttingen, Germany 1993.
5.
6.